Advertisements
Advertisements
प्रश्न
Write a chemical equation to illustrate the use of aluminium for joining cracked railway lines.
उत्तर
Aluminium is more reactive than iron and can be used for reducing iron oxide to iron. The equation for the same is: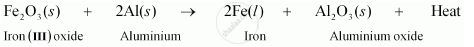
APPEARS IN
संबंधित प्रश्न
Metal A has electronic configuration (2, 8, 1) and metal B has (2, 8, 8, 2). Which is more reactive? Why? Identify these metals.
An element reacts with oxygen to give a compound with a high melting point. This compound is also soluble in water. The element is likely to be:
What changes in the colour of iron nails and copper sulphate solution do you observe after keeping the iron nails dipped in copper sulphate solution for about 30 minutes?
Give reason for the following:
Blue colour of copper sulphate solution is destroyed when iron filings are added to it.
Fill in the following blank with suitable word:
Magnesium liberates ............... gas on reacting with hot boiling water.
Explain why, metals usually do not liberate hydrogen gas with dilute nitric acid.
Write the equation for the reaction of Zinc with dilute hydrochloric acid.
Name the products formed. Also indicate the physical states of all the substances involved.
An acidic oxide is produced by the element:
(a) Na
(b) C
(c) Ca
(d) H
A metal less reactive and another metal more reactive than hydrogen are:
(a) aluminium and lead
(b) iron and magnesium
(c) copper and tin
(d) copper and mercury
What is the characteristic of the electronic configuration of noble gases?
