Advertisements
Advertisements
प्रश्न
Write a chemical equation to illustrate the use of aluminium for joining cracked railway lines.
उत्तर
Aluminium is more reactive than iron and can be used for reducing iron oxide to iron. The equation for the same is: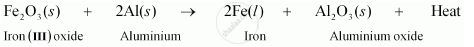
APPEARS IN
संबंधित प्रश्न
Metal A has electronic configuration (2, 8, 1) and metal B has (2, 8, 8, 2). Which is more reactive? Why? Identify these metals.
Fill in the following blank with suitable word:
Magnesium liberates ............... gas on reacting with hot boiling water.
Which of the following elements would yield an acidic oxide.
Na, S, C, K, H
Which of the following elements would yield a neutral oxide?
Na, S, C, K, H
What is the action of water on aluminium ? Write equation of the chemical reaction involved.
What special name is given to substances like aluminium oxide.
Write the equation for the reaction of Magnesium with dilute hydrochloric acid.
Name the products formed. Also indicate the physical states of all the substances involved.
You are given a solution of AgNO3. Which of the following do you think cannot displace Ag from AgNO3solution?
(a) Magnesium
(b) Zinc
(c) Gold
(d) Copper
The elements whose oxides can turn litmus solution red are:
(a) lithium and sodium
(b) copper and potassium
(c) carbon and hydrogen
(d) phosphorus and sulphur
Choose the correct alternative and rewrite the following:
Reaction of iron nails whith copper sulphate solution is an example of _____________________
