Advertisements
Advertisements
Question
Write a chemical equation to illustrate the use of aluminium for joining cracked railway lines.
Solution
Aluminium is more reactive than iron and can be used for reducing iron oxide to iron. The equation for the same is: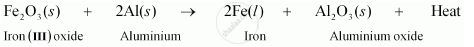
APPEARS IN
RELATED QUESTIONS
Give reasons why copper is used to make hot water tanks and not steel (an alloy of iron).
Why are metals called electropositive elements whereas non-metals are called electronegative elements?
Which of the following elements would yield a basic oxide.
Na, S, C, K, H
You are given samples of three metals − sodium, magnesium and copper. Suggest any two activities to arrange them in order of their decreasing reactivities.
What special name is given to substances like aluminium oxide.
Write the equation for the reaction of Zinc with dilute hydrochloric acid.
Name the products formed. Also indicate the physical states of all the substances involved.
What is meant by the reactivity series of metals? Arrange the following metals in an increasing order of their reactivities towards water:
Zinc, Iron, Magnesium, Sodium
In a solution of lead acetate, a strip of metal M was dipped. After some time, lead from the solution was deposited on the metal strip. Which metal is more reactive, M or lead?
In nature, metal A is found in a free state while metal B is found in the form of its compounds. Which of these two will be nearer to the top of the activity series of metals?
A zinc plate was kept in a glass container having CuSO4 solution. On examining it was found that the blue colour of the solution is getting lighter and lighter. After a few days, when the zinc plate was taken out of the solution, a number of small holes were noticed in it. State the reason and give chemical equation of the reaction involved.
