Advertisements
Advertisements
प्रश्न
Write the chemical reaction involved in Wolff-Kishner reduction.
Write the equations involved in the following reactions
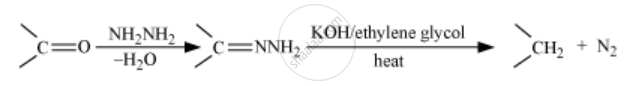
Wolff-Kishner reduction
उत्तर १
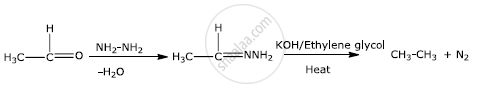
उत्तर २
Wolff-Kishner reduction:-
The Wolff Kishner reduction reaction is a method in which a carbonyl compound (aldehyde or ketone) is reduced to a hydrocarbon. In this method, the carbonyl compound is heated with a mixture of hydrazine and strong base such as sodium or potassium hydroxide at a temperature of 453 K - 473 K in ethylene glycol solvent.
APPEARS IN
संबंधित प्रश्न
Write the structures of the main products when acetone (CH3 − CO − CH3) reacts with the following reagent :
Zn − Hg/conc. HCl
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.
Zinc amalgam and dilute hydrochloric acid
How will you bring about the following conversion in not more than two steps?
Propanone to Propene
Complete the synthesis by giving missing starting material, reagent or product.
\[\ce{CH3COCH2COOC2H5 ->[(i) NaBH4][(ii) H+]}\]
Predict the products of the following reactions :
Predict the products of the following reactions :
Predict the main product of the following reaction:\[\begin{array}{c}
\ce{O\phantom{----}O\phantom{-}}\\
\ce{||\phantom{----}||\phantom{-}}\\
\ce{CH3-C-CH2-C-OCH3}
\end{array}\ce{->[(i)NaBH4][(ii)H+]}\]
Explain the following reactions:
Wolff Kishner reduction
Explain the following reactions:
Clemmensen reaction
Write the main product in the following reaction:
\[\ce{CH3CH2CHO ->[Zn(Hg)/Conc. HCl]}\]
