Advertisements
Advertisements
Questions
Write the chemical reaction involved in Wolff-Kishner reduction.
Write the equations involved in the following reactions
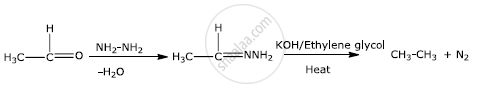
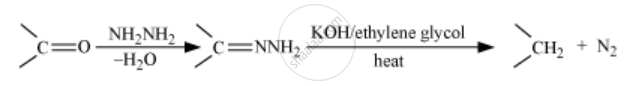
Wolff-Kishner reduction
Solution 1
Solution 2
Wolff-Kishner reduction:-
The Wolff Kishner reduction reaction is a method in which a carbonyl compound (aldehyde or ketone) is reduced to a hydrocarbon. In this method, the carbonyl compound is heated with a mixture of hydrazine and strong base such as sodium or potassium hydroxide at a temperature of 453 K - 473 K in ethylene glycol solvent.
APPEARS IN
RELATED QUESTIONS
Predict the products of the following reactions :
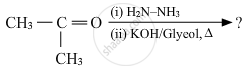
Write the structures of the main products when acetone (CH3 − CO − CH3) reacts with the following reagent :
Zn − Hg/conc. HCl
Complete the synthesis by giving missing starting material, reagent or product.
\[\ce{CH3COCH2COOC2H5 ->[(i) NaBH4][(ii) H+]}\]
Predict the products of the following reactions :
Predict the main product of the following reaction:\[\begin{array}{c}
\ce{O\phantom{----}O\phantom{-}}\\
\ce{||\phantom{----}||\phantom{-}}\\
\ce{CH3-C-CH2-C-OCH3}
\end{array}\ce{->[(i)NaBH4][(ii)H+]}\]
Write chemical equation of the following reaction :
Acetophenone is treated with `("Zn"("Hg"))/"Conc.HCl"`.
Reduction of > C = O to CH2 can be carried out with
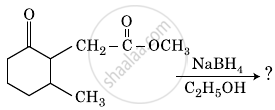
The product formed in the following chemical reaction is:
Explain the following reactions:
Wolff Kishner reduction
Explain the following reactions:
Clemmensen reaction
