Advertisements
Advertisements
Question
Write chemical equation of the following reaction :
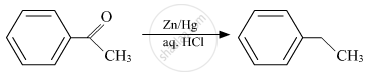
Acetophenone is treated with `("Zn"("Hg"))/"Conc.HCl"`.
Solution
The reaction of aldehydes and ketones with zinc amalgam (Zn/Hg) in concentrated HCl, reduces the aldehyde or ketone to a hydrocarbon and is called Clemmensen reduction.
RELATED QUESTIONS
How will you bring about the following conversions?
Propanone to propane
Write the chemical reaction involved in Wolff-Kishner reduction.
Predict the products of the following reactions :
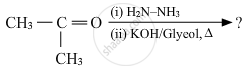
Write the structures of the main products when acetone (CH3 − CO − CH3) reacts with the following reagent :
Zn − Hg/conc. HCl
Predict the products of the following reactions :
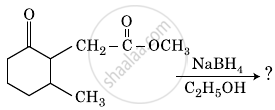
Predict the main product of the following reaction:\[\begin{array}{c}
\ce{O\phantom{----}O\phantom{-}}\\
\ce{||\phantom{----}||\phantom{-}}\\
\ce{CH3-C-CH2-C-OCH3}
\end{array}\ce{->[(i)NaBH4][(ii)H+]}\]
The product formed in the following chemical reaction is:
Explain the following reactions:
Wolff Kishner reduction
Explain the following reactions:
Clemmensen reaction
Write the main product in the following reaction:
\[\ce{CH3CH2CHO ->[Zn(Hg)/Conc. HCl]}\]
