Advertisements
Advertisements
प्रश्न
Write down the IUPAC name of the following complex and indicate the oxidation state, electronic configuration and coordination number. Also, give the stereochemistry and magnetic moment of the complex:
Cs[FeCl4]
उत्तर
| IUPAC name of the hybrid compound | Oxidation state of the central metal | Coordination number | Shape | Central metal ion | Electronic configuration | The value of n and the magnetic moment | Stereochemistry |
| Caesium tetrachloridoferrate(III) | +1 + x + 4 × (−1) = 0 ∴ x = +3 |
4 | Tetrahedral | Fe3+ | 3d5, \[\ce{t^3_{2g}e^2_g}\] | n = 5, `sqrt(5(5 + 2))` = 5.92 BM |
Optically inactive |
APPEARS IN
संबंधित प्रश्न
Write the structure and IUPAC names of isomeric aldehydes having molecular formula C5H10O.
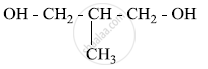
Write the IUPAC name of the given compound:
Write down the IUPAC name of the following complex: [Co(NH3)5 (NO2)](NO3)2
Write down the IUPAC name of the following complex: [Cr(NH3)2Cl2(en)]Cl (en = ethylenediamine)
Specify the oxidation number of the metal in the following coordination entity:
[Co(H2O)(CN)(en)2]2+
Specify the oxidation number of the metal in the following coordination entity:
[Cr(NH3)3Cl3]
Using IUPAC norms write the systematic name of the following:
[Pt(NH3)2Cl(NH2CH3)]Cl
Using IUPAC norms, write the systematic name of the following:
[Ti(H2O)6]3+
Using IUPAC norms, write the systematic name of the following:
[Mn(H2O)6]2+
Using IUPAC norms, write the systematic name of the following:
[NiCl4]2−
Using IUPAC norms, write the systematic name of the following:
[Ni(NH3)6]Cl2
Write down the IUPAC name of the following complex and indicate the oxidation state, electronic configuration and coordination number. Also, give the stereochemistry and magnetic moment of the complex:
[CrCl3(py)3]
The oxidation number of cobalt in K[Co(CO)4] is
(i) +1
(ii) +3
(iii) −1
(iv) −3
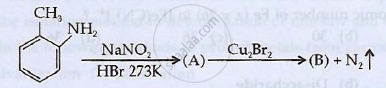
Identify 'A' and 'B' and rewrite the reactions
Write the IUPAC name of the following coordination compound:
K3[Fe(CN)6]
Write the IUPAC names of the following coordination compounds:
[CoBr2(en)2]+, (en = ethylenediamine)
Write the IUPAC name of the compound
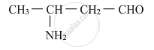
Write the IUPAC name of the K3[Fe(C2O4)3].
