Advertisements
Advertisements
प्रश्न
Write Lewis symbols for the following atoms and ions H and H–.
उत्तर १
H and H–
The number of valence electrons in hydrogen is 1.
The Lewis dot symbol of hydrogen (H) is 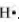
The uninegative charge infers that there will be one electron more in addition to the one valence electron. Hence, the Lewis dot symbol is 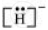
उत्तर २
`""_1"H" = 1` ∴ Lewis symbol = 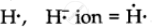
APPEARS IN
संबंधित प्रश्न
Write Lewis symbols for the following atoms and ions: S and S2–.
Write Lewis symbols for the following atoms and ions: Al and Al3+.
Use Lewis symbols to show electron transfer between the following atoms to form cations and anions:-
K and S
Draw Lewis dot diagram for the following.
Water (H2O)
Draw Lewis dot diagram for the following.
Carbon dioxide (CO2)
Draw Lewis dot diagram for the following.
Methane (CH4)
Draw Lewis electron dot structure of HF
Draw Lewis electron dot structure of SO2
Explain in brief with one example of an ionic bond.
Explain in brief with one example of a covalent bond
Explain in brief with one example of coordinate bond
____________ is an example of molecule with expanded octet.
Which of the following is INCORRECT with respect to enthalpy?
Which of the following molecule does not obey octet rule?
Which of the following molecule does not obey octet rule?
Match the items given in Column I with examples given in Column II.
| Column I | Column II |
| (i) Hydrogen bond | (a) \[\ce{C}\] |
| (ii) Resonance | (b) \[\ce{LiF}\] |
| (iii) Ionic solid | (c) \[\ce{H2}\] |
| (iv) Covalent solid | (d) \[\ce{HF}\] |
| (e) \[\ce{O3}\] |
Assertion (A): Sodium chloride formed by the action of chlorine gas on sodium metal is a stable compound.
Reason (R): This is because sodium and chloride ions acquire octet in sodium chloride formation.
