Advertisements
Advertisements
Question
Write Lewis symbols for the following atoms and ions H and H–.
Solution 1
H and H–
The number of valence electrons in hydrogen is 1.
The Lewis dot symbol of hydrogen (H) is 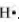
The uninegative charge infers that there will be one electron more in addition to the one valence electron. Hence, the Lewis dot symbol is 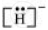
Solution 2
`""_1"H" = 1` ∴ Lewis symbol = 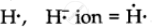
APPEARS IN
RELATED QUESTIONS
Explain the Formation of a Chemical Bond.
Write Lewis symbols for the following atoms and ions: Al and Al3+.
Use Lewis symbols to show electron transfer between the following atoms to form cations and anions:
Ca and O
Use Lewis symbols to show electron transfer between the following atoms to form cations and anions:
Al and N
Draw Lewis dot diagram for the following.
Hydrogen (H2)
Draw Lewis dot diagram for the following.
Water (H2O)
Draw Lewis dot diagram for the following.
Methane (CH4)
Draw Lewis dot diagram for the following.
Lifthium Fluoride (LiF)
Draw Lewis electron dot structure of HF
Draw Lewis electron dot structure of C2H4
Draw Lewis electron dot structure of CF3Cl
Draw Lewis electron dot structure of SO2
Explain in brief with one example of a covalent bond
Explain in brief with one example of coordinate bond
In which of the following, the central atom does NOT have lone pair(s) of electrons?
Which of the following molecule does not obey octet rule?
What is the formal charge on hydrogen atom in water molecule?
Match the items given in Column I with examples given in Column II.
| Column I | Column II |
| (i) Hydrogen bond | (a) \[\ce{C}\] |
| (ii) Resonance | (b) \[\ce{LiF}\] |
| (iii) Ionic solid | (c) \[\ce{H2}\] |
| (iv) Covalent solid | (d) \[\ce{HF}\] |
| (e) \[\ce{O3}\] |
