Advertisements
Advertisements
प्रश्न
Write the chemical equation for the following:
Combustion of methane
Write the equation for the complete combustion of:
- methane
- ethane
उत्तर
- \[\ce{CH4 + 2O2 -> CO2 + 2H2O}\]
- \[\ce{2C2H6 + 7O2 -> 4CO2 + 6H2O}\]
APPEARS IN
संबंधित प्रश्न
A student adds a spoon full of powdered sodium hydrogen carbonate to a flask containing ethanoic acid. List two main observations, he must note in his note book, about the reaction that takes place. Also write chemical equation foe the reaction.
A student adds 2 mL of acetic acid to a test tube containing 2 mL of distilled water. He then shakes the test tube well and leaves it to settle for some time. After about 5 minutes he observes that in the test tube there is :
(A) a clear transparent colourless solution
(B) a clear transparent pink solution
(C) a precipitate settling at the bottom of the test tube
(D) a layer of water the layer of acetic acid
Give the common names and IUPAC names of the following compounds of CH3COOH.
What happens when ethanoic acid reacts with sodium carbonate? Write chemical equation of the reaction involved.
What is an oxidising agent?
If you take a pinch of sodium hydrogen carbonate powder in a test-tube and add drop-by-drop acetic acid to it, what would you observe immediately? List any two main observations.
Which one of the following are the correct observations about acetic acid?
(A) It turns blue litmus red and smells like vinegar
(B) It turns blue litmus red and smells like burning sulphur
(C) It turns res litmus blue and smells like vinegar
(D) It turns red litmus blue and has a fruity smell
Acetic acid smells like:
(1) a banana
(2) vinegar
(3) an orange
(4) a lemon
How is ethyne prepared in the laboratory?
What is vinegar and glacial acetic acid?
What is the main constituent of vinegar?
Write a fully balanced equation for the folowing case:
Acitic acid is warmed with ethanol in the presence of concentrated sulpheric acid.
State how the following conversions can be carried out:
Ethyl alcohol to Ethane
Give reason for the following :
Ethyne is more reactive than ethane
Give balanced chemical equations for the following conversion :
Ethanoic acid to ethyl ethanote
Convert ethane to acetic acid.
Ethanoic acid turns ______ litmus to ______
Which of the following represents saponification reaction?
Observe the diagram given below and answer the questions:
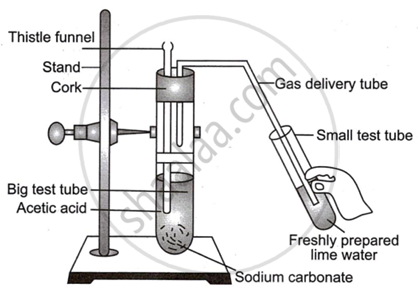
- Name the reactants in this reaction.
- Which gas comes out as effervescence in the bigger test tube?
- What is the colour change in the lime water?
- In the above experiment instead of sodium carbonate which chemical can be used to get same products?
- Write the use of acetic acid.
Give the balanced chemical equation of the reaction.
Oxidation of ethanol by acidified potassium dichromate.
