Advertisements
Advertisements
प्रश्न
Write the formula of the product formed when the element A (atomic number 19) combines with the element B (atomic number 17). Draw its electronic dot structure. What is the nature of the bond formed?
उत्तर
Element A has atomic number 19 so, it is potassium. Element B has atomic number 17 so, it is chlorine. As the valency of potassium is 1 and the valency of chlorine is also 1, the formula of the compound formed would be KCl or AB.
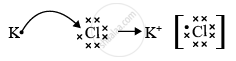
The nature of the bond between potassium and chlorine in KCl is an ionic bond.
APPEARS IN
संबंधित प्रश्न
Answer the following in respect of element `31/15 P `
Is it a reducing agent or oxidizing agent?
Arrange the elements of second and third periods in increasing order of their atomic size (excluding noble gases).
Why is the size of neon greater than fluorine?
Size of atom progressively becomes smaller when we move from sodium (Na) to chlorine (CI) in the third period of the periodic table?
Write the name and symbol of the element from the description.
The atom having the smallest atomic mass.
Write scientific reason.
Atomic radius goes on increasing down a group.
Atomic radius is expressed in the unit _______.
An element X has mass number 40 and contains 21 neutrons in its atom. To which group of the Periodic Table does it belong?
Which of the following gives the correct increasing order of the atomic radii of O, F and N?
- In below ladder symbols of elements are jumbled up. Rearrange these symbols of elements in the increasing order of their atomic number in the Periodic Table.
- Arrange them in the order of their group also.

