Advertisements
Online Mock Tests
Chapters
2: Solutions
3: Ionic Equilibria
4: Chemical Thermodynamics
5: Electrochemistry
6: Chemical Kinetics
7: Elements of Groups 16, 17 and 18
8: Transition and Inner transition Elements
9: Coordination Compounds
10: Halogen Derivatives
11: Alcohols, Phenols and Ethers
12: Aldehydes, Ketones and Carboxylic acids
13: Amines
14: Biomolecules
▶ 15: Introduction to Polymer Chemistry
16: Green Chemistry and Nanochemistry
![Balbharati solutions for Chemistry [English] 12 Standard HSC chapter 15 - Introduction to Polymer Chemistry Balbharati solutions for Chemistry [English] 12 Standard HSC chapter 15 - Introduction to Polymer Chemistry - Shaalaa.com](/images/chemistry-english-12-standard-hsc_6:74400b801d4c44ef8e058ff9d9dfe964.jpg)
Advertisements
Solutions for Chapter 15: Introduction to Polymer Chemistry
Below listed, you can find solutions for Chapter 15 of Maharashtra State Board Balbharati for Chemistry [English] 12 Standard HSC.
Balbharati solutions for Chemistry [English] 12 Standard HSC 15 Introduction to Polymer Chemistry Exercises [Pages 337 - 339]
Nylon fibres are ______.
semisynthetic fibres
polyamide fibres
polyester fibres
cellulose fibres
Choose the correct option from the given alternatives.
Which of the following is naturally occurring polymer?
Teflon
Polyethylene
PVC
Protein
Silk is a kind of _____ fibre.
Semisynthetic
Synthetic
Animal
Vegetable
Choose the correct option from the given alternatives.
Dacron is another name of ______.
Nylon 6
Orlon
Novolac
Terylene
Choose the correct option from the given alternatives.
Which of the following is made up of polyamides?
Dacron
Rayon
Nylon
Jute
Choose the correct option from the given alternatives.
The number of carbon atoms present in the ring of ɛ-caprolactam is ______.
Five
Two
Seven
Six
Terylene is _____.
polyamide fibre
polyester fibre
vegetable fibre
protein fibre
Choose the correct option from the given alternatives.
PET is formed by _______.
addition
condensation
alkylation
hydration
Choose the correct option from the given alternatives.
Chemically pure cotton is _______.
acetate rayon
viscose rayon
cellulose nitrate
cellulose
Choose the correct option from the given alternatives.
Teflon is chemically inert, due to presence of _______.
C-H bond
C-F bond
H- bond
C=C bond
Answer the following in one sentence.
Identify 'A' in the following reaction:
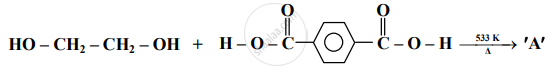
Answer the following in one sentence.
Identify 'B' in the following reaction:
\[\ce{H2N -(CH2)6 - NH2 + HOOC - (CH2)4 - COOH ->[N2][533 K]}\]'B'
Complete the following statement.
Caprolactam is used to prepare _______.
Complete the following statement.
Novolac is a copolymer of _______ and _______.
Complete the following statement.
Terylene is _______ polymer of terephthalic acid and ethylene glycol
Complete the following statement.
Benzoyl peroxide used in addition polymerisation acts as _______.
Complete the following statement.
Polythene consists of polymerized_______.
Draw the flow chart diagram to show the classification of polymers based on the type of polymerization.
Answer the following in one sentence.
Write examples of addition polymers and condensation polymers.
Answer the following in one sentence.
Name some chain-growth polymers.
Answer the following in one sentence.
Define the term: Monomer
Answer the following in one sentence.
Define the term: Vulcanization
Answer the following in one sentence.
Define the term: Synthetic fibres
Answer the following in one sentence.
What type of intermolecular force leads to high-density polymer?
Answer the following in one sentence.
Give one example each of copolymer and homopolymer.
Answer the following in one sentence.
Identify thermoplastic and thermosetting plastic from the following:
- PET
- Urea formaldehyde resin
- Polythene
- Phenol formaldehyde resin
Answer the following.
Write the names of classes of polymers formed according to intermolecular forces and describe briefly their structural characteristics.
Answer the following.
Write the reaction of the formation of Nylon 6.
Answer the following.
Write the reaction of the formation of Terylene.
Answer the following.
Write structure of natural rubber and neoprene rubber along with the name and structure of their monomers.
Answer the following.
Name the polymer type in which following linkage is present.
\[\begin{array}{cc}\ce{- C - O -}\\||\phantom{.....}\\
\ce{O\phantom{.....}}\end{array}\]
Answer the following.
Write structural formula of the following synthetic rubbers:
SBR rubber
Answer the following.
Write structural formula of the following synthetic rubbers:
Buna-N rubber
Answer the following.
Write structural formula of the following synthetic rubbers:
Neoprene rubber
Answer the following.
Match the following pairs:
| Name of polymer | Monomer |
| 1. Teflon | a. CH2 = CH2 |
| 2. PVC | b. CF2 = CF2 |
| 3. Polyester | c. CH2 = CHCl |
| 4. Polythene | d. C6H5OH and HCHO |
| 5. Bakelite | e. Dicarboxylic acid and polyhydoxyglycol |
Draw the structures of polymers formed from the following monomers
\[\ce{n HOOC–R–COOH + n HO–R'–OH}\]
Answer the following.
Draw the structures of polymers formed from the following monomers
H2N–(CH2)5 – COOH
Name and draw structure of the repeating unit in natural rubber.
Answer the following.
Classify the following polymers as natural and synthetic polymers
- Cellulose
- Polystyrene
- Terylene
- Starch
- Protein
- Silicones
- Orlon (Polyacrylonitrle)
- Phenol-formedehyde resins
Answer the following.
What are synthetic resins? Name some natural and synthetic resins.
Answer the following.
Distinguish between thermosetting and thermoplastic resins. Write example of both the classes.
Answer the following.
Write name and formula of raw material from which bakelite is made.
Identify condensation polymers and addition polymers from the following.
\[\begin{array}{cc}\ce{-(CH2 - CH -)_{n}}\\
\phantom{....}|\\\ce{\phantom{.......}C6H5}
\end{array}\]
Identify condensation polymers and addition polymers from the following.
-(CH2 - CH = CH - CH2 -)n
Identify condensation polymers and addition polymers from the following.
-(CO(CH2)4 - CONH(CH2)6NH-)n
Identify condensation polymers and addition polymers from the following.
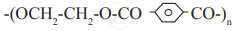
Attempt the following:
Write the chemical reactions involved in the manufacture of Nylon 6,6.
Attempt the following:
Explain the vulcanisation of rubber. Which vulcanizing agents are used for the following synthetic rubber?
a. Neoprene
b. Buna-N
Write chemical reaction to prepare the following polymer: Teflon
Write the reaction involved in the formation of:
Bakelite
Attempt the following:
What is meant by LDP and HDP? Mention the basic difference between the same with suitable examples.
Attempt the following:
Write preparation, properties and uses of Teflon.
Attempt the following:
Classify the following polymer as straight-chain, branched-chain and cross-linked polymers.
\[\begin{array}{cc}\ce{- (CH2 - CH -)_\text{n}}\\
\phantom{.....}|\\\ce{\phantom{.......}CN}\end{array}\]
Attempt the following:
Classify the following polymer as straight-chain, branched-chain and cross-linked polymers.
\[\begin{array}{cc}
\ce{-(CH2 - CH2 - CH - CH2 - CH2)_\text{n}}\\
|\phantom{..}\\\ce{\phantom{..}CH2}\\
|\phantom{..}\\\ce{\phantom{..}CH2}\end{array}\]
Attempt the following:
Classify the following polymer as straight-chain, branched-chain and cross-linked polymers.

Answer the following
How is polythene manufactured? Give their properties and uses.
Answer the following.
Is synthetic rubber better than natural rubber? If so, in what respect?
Answer the following.
Write main specialities of Buna-S, Neoprene rubber?
Write the structure of isoprene and the polymer obtained from it.
Answer the following.
Explain in detail free radical mechanism involved during preparation of addition polymer.
Solutions for 15: Introduction to Polymer Chemistry
![Balbharati solutions for Chemistry [English] 12 Standard HSC chapter 15 - Introduction to Polymer Chemistry Balbharati solutions for Chemistry [English] 12 Standard HSC chapter 15 - Introduction to Polymer Chemistry - Shaalaa.com](/images/chemistry-english-12-standard-hsc_6:74400b801d4c44ef8e058ff9d9dfe964.jpg)
Balbharati solutions for Chemistry [English] 12 Standard HSC chapter 15 - Introduction to Polymer Chemistry
Shaalaa.com has the Maharashtra State Board Mathematics Chemistry [English] 12 Standard HSC Maharashtra State Board solutions in a manner that help students grasp basic concepts better and faster. The detailed, step-by-step solutions will help you understand the concepts better and clarify any confusion. Balbharati solutions for Mathematics Chemistry [English] 12 Standard HSC Maharashtra State Board 15 (Introduction to Polymer Chemistry) include all questions with answers and detailed explanations. This will clear students' doubts about questions and improve their application skills while preparing for board exams.
Further, we at Shaalaa.com provide such solutions so students can prepare for written exams. Balbharati textbook solutions can be a core help for self-study and provide excellent self-help guidance for students.
Concepts covered in Chemistry [English] 12 Standard HSC chapter 15 Introduction to Polymer Chemistry are Introduction to Polymer Chemistry, Classification of Polymers, Some Important Polymers, Molecular Mass and Degree of Polymerization of Polymers, Biodegradable Polymers, Commercially Important Polymers, Preparation of Polytetrafluoroethene (Teflon).
Using Balbharati Chemistry [English] 12 Standard HSC solutions Introduction to Polymer Chemistry exercise by students is an easy way to prepare for the exams, as they involve solutions arranged chapter-wise and also page-wise. The questions involved in Balbharati Solutions are essential questions that can be asked in the final exam. Maximum Maharashtra State Board Chemistry [English] 12 Standard HSC students prefer Balbharati Textbook Solutions to score more in exams.
Get the free view of Chapter 15, Introduction to Polymer Chemistry Chemistry [English] 12 Standard HSC additional questions for Mathematics Chemistry [English] 12 Standard HSC Maharashtra State Board, and you can use Shaalaa.com to keep it handy for your exam preparation.
