Advertisements
Advertisements
प्रश्न
A certain gas 'X' occupies a volume of 100 cm3 at S.T.P. and weighs 0.5 g. Find its relative molecular mass.
उत्तर
At STP 100 cm3 of gas weights = 0.5 g
1 cm3 of a gas will weigh = `0.5/100`g
∴ At STP 22400 cm3 of gas will weight = `0.5/100 xx 22400`
= 112 g
APPEARS IN
संबंधित प्रश्न
Give balanced chemical equations for the following conversions A, B, and C:
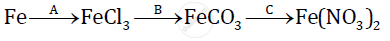
The usefulness of a fertilizer depends upon percentage of nitrogen present in it. Find which of the following is a better fertilizer:
(a) Ammonium nitrate [NH4NO3]
(b) Ammonium phosphate [(NH4)3PO4 (N=14,H=1,O=16,P=31)
What is the mass of nitrogen in 1000Kg of urea [CO(NH2)2] ?
[H = 1, C= 12, N= 14, O = 16]
The reaction of potassium permanganate (VII) with acidified iron (II) sulphate is given below:
2KMno4 + 10FeSO4 + 8H2O → K2SO4 + 2MnSO4 + 5Fe2(SO4)3 + 8H2O
If 15.8g of potassium permanganate (VII) was used in the reaction, calculate the mass of iron (II) sulphate used in the above reaction.
When heated, potassium permanganate decomposes according to the following equation:
\[\ce{2KMnO4 -> \underset{solid residue}{K2MnO4 + MnO2} + O2}\]
Given that the molecular mass of potassium permanganate is 158 g, what volume of oxygen (measured at room temperature) would be obtained by the complete decomposition of 15.8 g of potassium permanganate? (Molar volume at room temperature is 24 litres). [K = 39, Mn = 55, O = 16]
Calculate the percentage of nitrogen in aluminium nitride. [Al = 27, N = 14]
Calculate the relative molecular mass of:
Ammonium chloroplatinate (NH4)2 PtCl6
Calculate the relative molecular mass of:
Potassium chlorate
Find the weight of 0.5 mole of O2.
Calculate the volume occupied at S.T.P. by 2 moles of SO2.
