Advertisements
Advertisements
प्रश्न
A first order reaction is 50% complete in 25 minutes. Calculate the time for 80% completion of the reaction.
उत्तर
`t_{1//2} = 25 ` minutes ⇒k = `0.693 /{25 "mins"}`
K = `2.303/25 = 2.303/tlog 100/{a_1} `
⇒ `0.693/25 = 2.303/t log 100/20` 80% completion
`t = 2.303/0.693 xx 25 xx 0.6990`
t = 57.3 mins
APPEARS IN
संबंधित प्रश्न
Time required to decompose SO2Cl2 to half of its initial amount is 60 minutes. If the decomposition is a first order reaction, calculate the rate constant of the reaction.
The time required for 10% completion of a first order reaction at 298 K is equal to that required for its 25% completion at 308 K. If the value of A is 4 × 1010 s−1. Calculate k at 318 K and Ea.
Which of the following graphs is correct for a first order reaction?

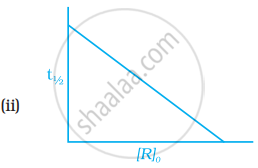
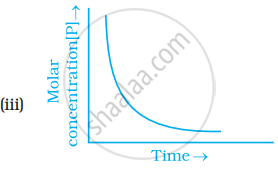
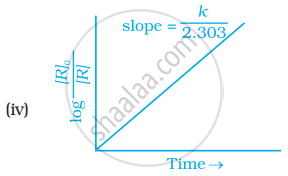
First order reaction is 50% complete in 1.26 × 1014s. How much time could it take for 100% completion?
In the first order reaction, half of the reaction is complete in 100 seconds. The time for 99% of the reaction to occurs will be
The reaction X → product
Follow first order of kinetics. In 40 minutes the concentration of 'X' changes from 0.1 m to 0.025. M. The rate of reaction when concentration of X is 0.01 m is.
In a first order reaction the concentration of reactants decreases from 400mol L-1 to 25 mol L-1 in 200 seconds. The rate constant for the reaction is ______.
Radioactive decay follows first-order kinetics. The initial amount of two radioactive elements X and Y is 1 gm each. What will be the ratio of X and Y after two days if their half-lives are 12 hours and 16 hours respectively?
Slove: \[\ce{2NOBr -> 2NO_{2(g)} + Br_{2(g)}}\]
For the above reaction, the rate law is rate = k[NOBr]2. If the rate of reaction is 6.5 × 10−6 mol L−1 s−1 at 2 × 10−3 mol L−1 concentration of NOBr, calculate the rate constant k for the reaction.
Show that `t_(1/2)= 0.693/k` for first reaction.
