Advertisements
Advertisements
प्रश्न
A student performs an experiment on photoelectric effect, using two materials A and B. A plot of Vstop vs ν is given in Figure.
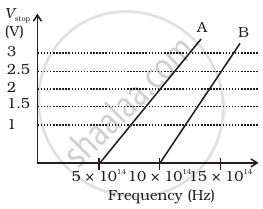
- Which material A or B has a higher work function?
- Given the electric charge of an electron = 1.6 × 10–19 C, find the value of h obtained from the experiment for both A and B.
Comment on whether it is consistent with Einstein’s theory:
उत्तर
Threshold frequency (no): The minimum frequency of incident radiations, required to eject the electron from the metal surface is defined as the threshold frequency.
If incident frequency n < n0 ⇒ No photoelectron emission.
For most metals, the threshold frequency is in the ultraviolet (corresponding to wavelengths between 200 and 300 nm), but for potassium and caesium oxides it is in the visible spectrum (A between 400 and 700 nm).
Here we are given the threshold frequency of A
i. vOA = 5 × 1014 Hz and
For B, vOB = 10 × 014 Hz
We know that
Work function, `phi = hv_0` or `phi_0 ∝ v_0`
⇒ `phi_0 ∝ v_0`
So, `phi_(OA)/phi_(OB) = (5 xx 10^14)/(10 xx 10^14) < 1`
⇒ `phi_(OA) < phi_(OB)`
Thus, work function of B is higher than A.
ii. For metal A, slope = `h/e = 2/(10 - 15)10^14`
or `h = (2e)/(5 xx 10^14) = (2 xx 1.6 xx 10^-19)/(5 xx 10^14)`
= `6.4 xx 10^-34` Js
For metal B, sloe = `h/e = 2.5/((15 - 10)10^14`
or `h = (2.5 xx e)/(5 xx 10^14) = (2.5 xx 1.6 xx 10^-19)/(5 xx 10^14)`
= 8 × 10–34 Js
Since the value of h from the experiment for metals A and B is different. Hence, the experiment is not consistent with the theory.
APPEARS IN
संबंधित प्रश्न
The work function for a certain metal is 4.2 eV. Will this metal give photoelectric emission for incident radiation of wavelength 330 nm?
Plot a graph showing the variation of photoelectric current with collector plate potential at a given frequency but for two different intensities I1 and I2, where I2 > I1.
point out any two characteristic properties of photons on which Einstein’s photoelectric equation is based ?
Define the terms (i) ‘cut-off voltage’ and (ii) ‘threshold frequency’ in relation to the phenomenon of photoelectric effect.
Using Einstein’s photoelectric equation shows how the cut-off voltage and threshold frequency for a given photosensitive material can be determined with the help of a suitable plot/graph.
A monochromatic light source of intensity 5 mW emits 8 × 1015 photons per second. This light ejects photoelectrons from a metal surface. The stopping potential for this setup is 2.0 V. Calculate the work function of the metal.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
In a photoelectric experiment, the collector plate is at 2.0 V with respect to the emitter plate made of copper (φ = 4.5 eV). The emitter is illuminated by a source of monochromatic light of wavelength 200 nm. Find the minimum and maximum kinetic energy of the photoelectrons reaching the collector.
Consider the situation of the previous problem. Consider the faster electron emitted parallel to the large metal plate. Find the displacement of this electron parallel to its initial velocity before it strikes the large metal plate.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
The wavelength of a photon needed to remove a proton from a nucleus which is bound to the nucleus with 1 MeV energy is nearly ______.
There are materials which absorb photons of shorter wavelength and emit photons of longer wavelength. Can there be stable substances which absorb photons of larger wavelength and emit light of shorter wavelength.
The photon emitted during the de-excitation from the first excited level to the ground state of a hydrogen atom is used to irradiate a photocathode in which the stopping potential is 5 V. Calculate the work function of the cathode used.
