Advertisements
Advertisements
प्रश्न
The wavelength of a photon needed to remove a proton from a nucleus which is bound to the nucleus with 1 MeV energy is nearly ______.
पर्याय
1.2 nm
1.2 × 10–3 nm
1.2 × 10–6 nm
1.2 × 101 nm
उत्तर
The wavelength of a photon needed to remove a proton from a nucleus which is bound to the nucleus with 1 MeV energy is nearly `underline(1.2 xx 10^-3 nm)`.
Explanation:
According to Einstein’s quantum theory light propagates in the bundles (packets or quanta) of energy, each bundle is called a photon and possessing energy. Energy of photon is given by
`E = hv = (hc)/λ`; where c = Speed of light, h = Planck's constant = `6.6 xx 10^-34` J-sec, v = Frequency in Hz, λ = the minimum wavelength of the photon required to eject the proton from nucleus.
In electron volt, `E(eV) = (hc)/(eλ) = 12375/(λ(Å)) = 12400/(λ(Å))`
According to the problem,
Energy of a photon, E = 1 MeV or 106 eV
Now, hc = 1240 eV nm
Now, `E = (hc)/λ`
⇒ λ = `(hc)/E = 1240/10^6` nm
= 1.24 × 10–3 nm
APPEARS IN
संबंधित प्रश्न
In an experiment on the photoelectric effect, the slope of the cut-off voltage versus the frequency of incident light is found to be 4.12 × 10−15 Vs. Calculate the value of Planck’s constant.
Briefly explain the three observed features which can be explained by Einstein’s photoelectric equation.
The electric field at a point associated with a light wave is `E = (100 "Vm"^-1) sin [(3.0 xx 10^15 "s"^-1)t] sin [(6.0 xx 10^15 "s"^-1)t]`.If this light falls on a metal surface with a work function of 2.0 eV, what will be the maximum kinetic energy of the photoelectrons?
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
In a photoelectric experiment, the collector plate is at 2.0 V with respect to the emitter plate made of copper (φ = 4.5 eV). The emitter is illuminated by a source of monochromatic light of wavelength 200 nm. Find the minimum and maximum kinetic energy of the photoelectrons reaching the collector.
How does one explain the emission of electrons from a photosensitive surface with the help of Einstein’s photoelectric equation?
Choose the correct answer from given options
Photons of frequency v are incident on the surface of two metals A and B of threshold frequency 3/4 v and 2/3 v, respectively. The ratio of maximum kinetic energy of electrons emitted from A to that from B is
Each photon has the same speed but different ______.
A student performs an experiment on photoelectric effect, using two materials A and B. A plot of Vstop vs ν is given in Figure.
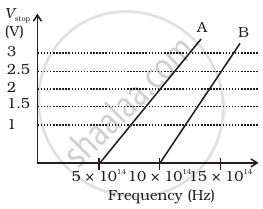
- Which material A or B has a higher work function?
- Given the electric charge of an electron = 1.6 × 10–19 C, find the value of h obtained from the experiment for both A and B.
Comment on whether it is consistent with Einstein’s theory:
Radiation of frequency 1015 Hz is incident on three photosensitive surfaces A, B and C. Following observations are recorded:
Surface A: no photoemission occurs
Surface B: photoemission occurs but the photoelectrons have zero kinetic energy.
Surface C: photo emission occurs and photoelectrons have some kinetic energy.
Using Einstein’s photo-electric equation, explain the three observations.
A photon of wavelength 663 nm is incident on a metal surface. The work function of the metal is 1.50 eV. The maximum kinetic energy of the emitted photoelectrons is ______.
