Advertisements
Advertisements
प्रश्न
Account for the following:
Sc3+ is colourless whereas Ti3+ is coloured in an aqueous solution.
Account for the following:
Ti3+ is coloured whereas Sc3+ is colourless in an aqueous solution.
उत्तर
Sc3+: [Ar]18
Ti3+: [Ar]18 3d1
Ti3+ is coloured in an aqueous solution due to the presence of one unpaired electron, whereas Sc3+ is colourless.
APPEARS IN
संबंधित प्रश्न
In 3d series (Sc to Zn), which element has the lowest enthalpy of atomisation and why?
Compare the general characteristics of the first series of the transition metals with those of the second and third series metals in the respective vertical columns. Give special emphasis on the following point:
Electronic configurations
An antifriction alloy made up of antimony with tin and copper, which is extensively used in machine bearings is called _______.
(A) Duralumin
(B) Babbitt metal
(C) Spiegeleisen
(D) Amalgam
Give reasons Iron has the higher enthalpy of atomization than that of copper.
The magnetic nature of elements depends on the presence of unpaired electrons. Identify the configuration of transition element, which shows highest magnetic moment.
Which of the following statements is not correct?
Assertion: Separation of \[\ce{Zr}\] and \[\ce{Hf}\] is difficult.
Reason: Because \[\ce{Zr}\] and \[\ce{Hf}\] lie in the same group of the periodic table.
It has been observed that first ionization energy of 5 d series of transition elements are higher than that of 3d and 4d series, explain why?
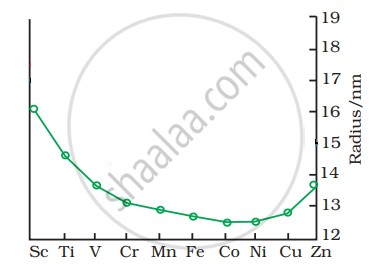
The trend of which property is represented by the following graph?
A coordination compound has the formula \[\ce{CoCl3.4NH3}\]. It precipitates silver ions as AgCl and its molar conductance corresponds to a total of two ions.
Based on this information, answer the following question:
- Deduce the structural formula of the complex compound.
- Write the IUPAC name of the complex compound.
- Draw the geometrical isomers of the complex compound.
