Advertisements
Advertisements
प्रश्न
An alcohol (x) gives blue colour in Victormeyer’s test and 3.7 g of X when treated with metallic sodium liberates 560 mL of hydrogen at 273 K and 1 atm pressure what will be the possible structure of X?
पर्याय
CH3CH(OH)CH2CH3
CH3 – CH(OH) – CH3
CH3 – C(OH) (CH3)2
CH3 – CH2 – CH(OH) – CH2 – CH3
उत्तर
CH3CH(OH)CH2CH3
Explanation:
\[\ce{2R - OH + 2Na -> 2RONa + H2 ^}\] 2 moles of alcohol gives 1 mole of H2 which occupies 22.4 L at 273 K and 1 atm
∴ number of moles of alcohol = `(2 "moles of R" - "OH")/(22.4 "L of H"_2) xx 560 "mL"`
= 0.05 moles
∴ no. of moles = `"mass"/"molar mass"`
molar mass = `3.7/0.05` = 74 g mol−1
General formula for R – OH Cn H2n+1 – OH
∴ n(12) + (2n+1) (1) + 16 + 1 = 74
14n = 74 – 18
14n = 56
∴ n = `56/14` = 4
The 2° alcohol which contains 4 carbon is CH3CH(OH)CH2CH3
APPEARS IN
संबंधित प्रश्न
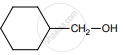 on treatment with Con. H2SO4 predominately gives
on treatment with Con. H2SO4 predominately gives
Carbolic acid is ____________.
In the reaction \[\ce{Ethanol ->[PCl5] X ->[alc. KOH] Y ->[H2SO4/H2O][298 K] Z}\]. The ‘Z’ is
Predict the major product, when 2-methyl-but-2-ene is converted into alcohol in the following method.
Hydroboration
Predict the major product, when 2-methyl-but-2-ene is converted into alcohol in the following method.
Hydroxylation using bayers reagent
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI
What will be the product (X and A)for the following reaction
acetylchloride \[\ce{{acetylchloride}->[i)CH3MgBr][ii)H3O+]X ->[acidK2Cr2O7] A}\]
Predict the major product, when 2-methyl but -2-ene is converted into an alcohol in each of the following method.
Acid catalysed hydration
What will be the product (X and A)for the following reaction
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O^+]X ->[acid K2Cr2O7]A}\]
