Advertisements
Advertisements
प्रश्न
An organic compound 'A' with molecular formula C5H8O2 is reduced to n-pentane with hydrazine followed by heating with NaOH and glycol. 'A' forms a dioxime with hydroxylamine and gives a positive iodoform and Tollen's test. Identify 'A' and give its reaction for iodoform and Tollen's test.
उत्तर
Molecular formula = C5H8O2 (given)
Structural formula:
\[\begin{array}{cc}
\phantom{...}\ce{O}\phantom{.............}\ce{O}\phantom{}\\
\phantom{...}||\phantom{..............}||\phantom{}\\
\ce{CH3-C-CH2-CH2-C-H}
\end{array}\]
Compound 'A' Hydrazine:
\[\ce{CH3-CH2-CH2-\underset{n-pentane}{CH2-CH3} + 2H2O}\]
\[\begin{array}{cc}
\phantom{...............}\ce{O}\phantom{.............}\ce{O}\phantom{....................}\ce{NOH}\phantom{..........}\ce{NOH}\phantom{.....................}\ce{O}\phantom{..............}\ce{O}\phantom{.............}\\
\phantom{.................}||\phantom{..............}||\phantom{....................}||\phantom{..............}||\phantom{.........................}||\phantom{..............}||\phantom{................}\\
\ce{CH3-C-CH2-CH2-C-H ->[2NH2OH]\underset{dioxime}{CH3-C-CH2}-CH2-C-H ->[Tollen's Reagent] CH3-C-CH2-CH2-C-OH}\\
\end{array}\]
\[\begin{array}{cc}
\ce{O}\phantom{........................................................}\\
||\phantom{........................................................}\\
\ce{CH3-C-CH2-CH2-CHO ->[NaOH][I2] NaOOC-CH2-CH2-CHO + \underset{Iodoforrn}{CHI3}}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
A and B are two functional isomers of compound C3H6O.On heating with NaOH and I2, isomer B forms yellow precipitate of iodoform whereas isomer A does not form any precipitate. Write the formulae of A and B.
Which sugar does not reduce Fehling's solution?
Which of the following tests/reactions is given by aldehydes as well as ketones?
A hydrocarbon (A) with molecular formula C5H10 on ozonolysis gives two products (B) and (C). Both (B) and (C) give a yellow precipitate when heated with iodine in presence of NaOH while only (B) give a silver mirror on reaction with Tollen’s reagent.
- Identify (A), (B) and (C).
- Write the reaction of B with Tollen’s reagent.
- Write the equation for iodoform test for C.
- Write down the equation for aldol condensation reaction of B and C.
Write chemical test to distinguish between the following compounds:
Phenol and Benzoic acid
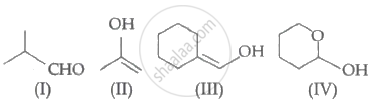
Which among the above compound/s does/do not form Silver mirror when treated with Tollen's reagent?
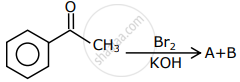
The major products formed in the following reaction sequence A and B are:
The correct set of products obtained in the following reactions:
- \[\ce{RCN ->[reduction]}\]
- \[\ce{RCN ->[(i) CH3MgBr][(ii) H2O]}\]
- \[\ce{RNC ->[hydrolysis]}\]
- \[\ce{RNH2 ->[HNO2]}\]
In Tollen's test for aldehyde, the overall number of electrons(s) transferred to the Tollen's reagent formula \[\ce{[Ag(NH3)2]+}\] per aldehyde group to form silver mirror is ______. (Round off to the nearest integer)
The reagent that can be used to distinguish acetophenone and benzophenone is ______.
