Advertisements
Advertisements
प्रश्न
Answer the following question :
Name a metal which is found abundantly in earth's crust.
उत्तर
A metal which is found abundantly in the Earth's crust is aluminium.
APPEARS IN
संबंधित प्रश्न
Write the equation for the reaction where the aluminum oxide for the electrolytic extraction of aluminum is obtained by heating aluminum hydroxide.
Name the following:
The chief ore of aluminium.
Name the following:
The materials used as electrodes in the electrolytic extraction of aluminium.
In the Hall's process for extraction of aluminium, Sate the location of cathode and anode and explain what occurs at each electrode.
What is the role of cryolite (NaAlF6) in the electrolytic reduction of alumina in the Hall's process?
Aluminium is extracted from its chief ore bauxite. The ore is first purified and then the metal is extracted from it by electrolytic reduction.
Write three balanced equations for the purification of bauxite by Hall's process.
Explain the reason:
Why 'food containing iron salts' should not be cooked in aluminium utensils?
Name the alloy used for the following purpose.
Surgical instruments
The following sketch illustrates the process of conversion of Alumina to Aluminium:
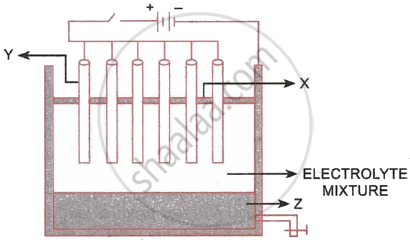
- Name the constituent of the electrolyte mixture which has a divalent metal in it.
- Name the powdered substances ‘X’ sprinkled on the surface of the electrolyte mixture.
- What is the name of the process?
- Write the reactions taking place at the electrodes ‘Y’ (anode) and ‘Z’ (cathode), respectively.
The compound that is not a constituent of the electrolytic mixture used in Hall-Heroult's process is ______.
