Advertisements
Advertisements
प्रश्न
The following sketch illustrates the process of conversion of Alumina to Aluminium:
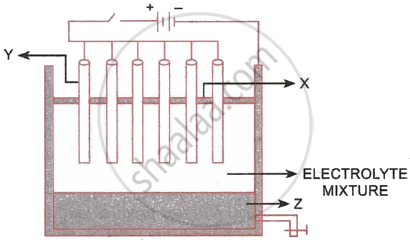
- Name the constituent of the electrolyte mixture which has a divalent metal in it.
- Name the powdered substances ‘X’ sprinkled on the surface of the electrolyte mixture.
- What is the name of the process?
- Write the reactions taking place at the electrodes ‘Y’ (anode) and ‘Z’ (cathode), respectively.
उत्तर
- The electrolytes used are molten alumina, cryolite and fluospar.
- Powdered coke layer (X).
- Hall Heroult process
- Cathode (Z): \[\ce{Al^{3+} + 3e^{-} -> Al}\]
Anode (Y): \[\ce{C + 2O^{2+} -> CO_2 + 4e^{-}}\]
APPEARS IN
संबंधित प्रश्न
Explain why In the electrolysis of alumina using the Hall Heroult's Process the electrolyte is covered with powdered coke
Aluminium is a more active metal than iron, but suffers less corrosion. Why?
Name the constituents of Bronze.
What is the role of cryolite (NaAlF6) in the electrolytic reduction of alumina in the Hall's process?
The following question relate to the extraction of aluminium by electrolysis.
Give the equation for the reaction that takes place at the cathode
Name the solution used to react with bauxite as a first step in obtaining pure aluminium oxide, in the Baeyer's process.
Explain the reason:
Why 'food containing iron salts' should not be cooked in aluminium utensils?
The chief ore of Aluminium is ______
Write the balanced chemical equation to show the concentration of ore in Baeyer’s process.
Sodium aluminate to aluminium hydroxide
State true or false.
Bauxite is the main ore of aluminium.
