Advertisements
Advertisements
प्रश्न
Name the constituents of Bronze.
उत्तर
The constituents of Bronze are copper (80%), tin (18%) and zinc (2%).
APPEARS IN
संबंधित प्रश्न
Name the following: A compound added to lower the fusion temperature of electrolytic bath in the extraction of aluminum
For graphite, explain its significance in the extraction of aluminium.
The reason for using Aluminium in the alloy duralumin is ______.
Name the following:
The chief ore of aluminium.
Give reason for the following:
Extraction of aluminium was very difficult in the beginning.
Aluminium is a more active metal than iron, but suffers less corrosion. Why?
For the substance listed below, explain its role in the extraction of aluminium: Cryolite
The following is an extract from 'Metals in the Service of Man, Alexander and Street/Pelican 1976':
| 'Alumina (aluminium oxide) has a very high melting point of over 2000°C so that it cannot readily be liquefied. However, conversion of alumina to aluminium and oxygen, by electrolysis, can occur when it is dissolved in some other substance.' |
- Which solution is used to react with bauxite as a first step in obtaining pure aluminium oxide?
- The aluminium oxide for the electrolytic extraction of aluminium is obtained by heating aluminium hydroxide. Write the balanced chemical equation for this reaction.
- Name the element which serves both as the anode and the cathode in the extraction of aluminium.
- Write the balanced chemical equation for the reaction that occurs at the cathode during the extraction of aluminium by electrolysis.
- Give the balanced chemical equation for the reaction which occurs at the anode when aluminium is purified by electrolysis.
The sketch below illustrates the refin ing of aluminium by Hoope's process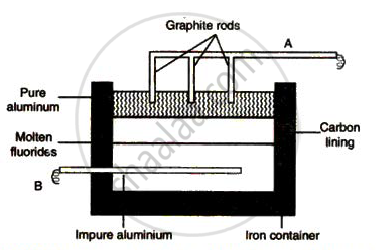
(a) Which of A and B is th e cathode and which one is the anode?
(b) What is the electroly te in the tank?
( c) What material is used for th e cathode?
Write the constituents of the electrolyte for the extraction of aluminium.
