Advertisements
Advertisements
प्रश्न
Write the balanced chemical equation to show the concentration of ore in Baeyer’s process.
Sodium aluminate to aluminium hydroxide
उत्तर
\[\ce{NaAlO2 + 2H2O -> NaOH + Al(OH)3 (↓)}\]
Explanation:
Sodium aluminate on hydrolysis at 50°C gives sodium hydroxide and precipitates aluminium hydroxide. This step is done in order to remove the impurities.
APPEARS IN
संबंधित प्रश्न
Aluminium is a more active metal than iron, but suffers less corrosion. Why?
Name the constituents of Bronze.
For the substance listed below, explain its role in the extraction of aluminium: Sodium hydroxide
For the substance listed below, explain its role in the extraction of aluminium: Graphite
Write the equation for the reaction where the aluminium oxide for the electrolytic extraction of aluminium is obtained by heating aluminium hydroxide.
During the concentration of bauxite ore, aluminium goes in ______ part because of its ______ nature.
Explain the reason:
Why 'food containing iron salts' should not be cooked in aluminium utensils?
Name the following :
The compound formed by the reaction between calcium oxide and silica.
Name the alloy used for the following purpose.
Making parts of watches
The following sketch illustrates the process of conversion of Alumina to Aluminium:
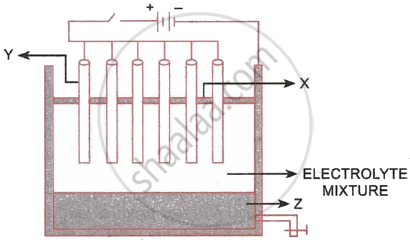
- Name the constituent of the electrolyte mixture which has a divalent metal in it.
- Name the powdered substances ‘X’ sprinkled on the surface of the electrolyte mixture.
- What is the name of the process?
- Write the reactions taking place at the electrodes ‘Y’ (anode) and ‘Z’ (cathode), respectively.
