Advertisements
Advertisements
प्रश्न
Answer the following question.
What type of stoichiometric defect is shown by ZnS and why?
उत्तर
The stoichiometric defect shown by ZnS is Frenkel defect which is due to the large difference in the size of the ions, i.e. due to the small size of Zn2+.
APPEARS IN
संबंधित प्रश्न
Give reasons : In stoichiometric defects, NaCl exhibits Schottky defect and not Frenkel defect.
Defect shown by non-ionic solids is ____________.
Type of stoichiometric defect shown by ZnS is ____________.
Type of stoichiometric defect shown by AgBr is ____________.
The following diagram shows:
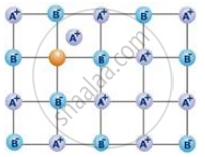
Schottky defect defines imperfection in the lattice structure of ____________.
Assertion: Due to Frenkel defect, there is no effect on the density of the crystalline solid.
Reason: In Frenkel defect, no cation or anion leaves the crystal.
Schottky defect is observed in crystals when ______.
Match the defects given in Column I with the statements in given Column II.
| Column I | Column II |
| (i) Simple vacancy defect | (a) shown by non-ionic solids and increases density of the solid. |
| (ii) Simple interstitial defect | (b) shown by ionic solids and decreases density of the solid. |
| (iii) Frenkel defect | (c) shown by non ionic solids and density of the solid decreases. |
| (iv) Schottky defect | (d) shown by ionic solids and density of the solid remains the same. |
Schottky defect is noticed in ______
