Advertisements
Advertisements
प्रश्न
Answer the following question.
Write one stereochemical difference between SN1 and SN2 reactions.
उत्तर
SN1 reactions lead to the formation of racemic mixture whereas SN2 reactions lead to inversion in the configuration of a product.
APPEARS IN
संबंधित प्रश्न
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH2Br + KCN ->[aq.ethanol]}\]
The stability order for carbocation is _______.
(A) 2° > 3° > 1°
(B) 3° > 2° > 1°
(C) 3° > 1° > 2°
(D) 1° > 3° > 2°
SN2 mechanism proceeds through intervention of ____________.
Identify the end product (C) in the following sequence:
\[\ce{C2H5OH ->[SOCl2][Pyridine] A ->[KCN {(alc.)}] B ->[2H2O/H^+] C}\]
The increasing order of nucleophilicity would be:
Complete the following analogy:
Same molecular formula but different structures: A : : Non superimposable mirror images: B
Aryl halides are extremely less reactive towards nucleophilic substitution. Predict and explain the order of reactivity of the following compounds towards nucleophilic substitution:
| (I) | 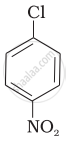 |
| (II) | 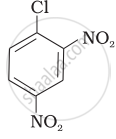 |
| (III) | 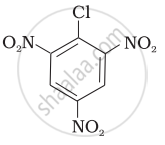 |
Which of the following is the definition of chirality?
Inversion of configuration occurs in ______.
Which alkyl halide from the following pair would you expect to react more rapidly by an SN2 mechanism? Explain your answer.
\[\begin{array}{cc}\ce{CH3CHCH2CH2Br}\\|\phantom{.........}\\\ce{CH3}\phantom{......}\end{array}\] or \[\begin{array}{cc}\ce{CH3CH2CHCH2Br}\\\phantom{}|\\\phantom{...}\ce{CH3}\end{array}\]
