Advertisements
Advertisements
प्रश्न
Arrange the following:
In increasing order of basic strength:
C6H5NH2, C6H5NHCH3, C6H5CH2NH2
उत्तर
C6H5NHCH3 is more basic than C6H5NH2 due to the presence of an electron-donating −CH3 group in C6H5NHCH3.
Again, in C6H5NHCH3, −C6H5 group is directly attached to the N-atom. However, it is not so in C6H5CH2NH2. Thus, in C6H5NHCH3, the −R effect of −C6H5 group decreases the electron density over the N-atom. Therefore, C6H5CH2NH2 is more basic than C6H5NHCH3.
Hence, the increasing order of the basic strengths of the given compounds is as follows:
C6H5NH2 < C6H5NHCH3 < C6H5CH2NH2
संबंधित प्रश्न
Arrange the following:
Aniline, p-nitroaniline, p-methylaniline - in the increasing order of their basic strength
Write the structures of main products when aniline reacts with the following reagents : HCl
Arrange the following:
In increasing order of basic strength:
C6H5NH2, C6H5N(CH3)2, (C2H5)2NH and CH3NH2
Arrange the following:
In increasing order of basic strength:
Aniline, p-nitroaniline and p-toluidine
Arrange the following:
In decreasing order of basic strength in the gas phase:
C2H5NH2, (C2H5)2NH, (C2H5)3N and NH3
Write a short note on the following:
Carbylamine reaction
Write the structures of the main products of the following reactions:
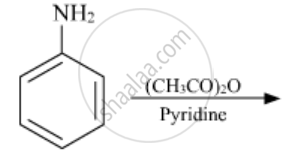
Write the structures of the main products of the following reactions:

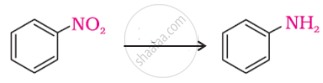
The following reaction takes place in the presence of:
Arrange the following in decreasing order of their basic strength:
C6H5NH2, C2H5NH2, (C2H5)2NH2, NH3
Explain why \[\ce{MeNH2}\] is stronger base than \[\ce{MeOH}\]?
When ethanol is mixed with ammonia and passed over alumina the compound formed is which compound?
Which of the following is most basic?
What is the correct decreasing order of the basic character of the three amines and ammonia?
Account for the following:
Arrange the following compounds in the increasing order of their basic strength in aqueous solution: CH3NH2,(CH3)3N,(CH3)2NH
Among the following, which has the highest value of pKb?
The correct order of the increasing basic nature of Ammonia, Methylamine and Aniline is:
