Advertisements
Advertisements
प्रश्न
Assertion: The boiling points of alkyl halides decrease in the order:
\[\ce{RI > RBr > RCl > RF}\]
Reason: The boiling points of alkyl chlorides, bromides and iodides are considerably higher than that of the hydrocarbon of comparable molecular mass.
पर्याय
Assertion and reason both are correct and reason is correct explanation of assertion.
Assertion and reason both are wrong statements.
Assertion is correct but reason is wrong statement.
Assertion is wrong but reason is correct statement.
Assertion and reason both are correct statements but reason is not correct explanation of assertion.
उत्तर
Assertion and reason both are correct statements but reason is not correct explanation of assertion.
Explanation:
The given sequence of boiling points is correct:
RI > RBr > RCl > RF
Halides are polar molecules hence they have greater boiling points than hydrocarbons.
APPEARS IN
संबंधित प्रश्न
Arrange the set of compounds in order of increasing boiling points.
Bromomethane, Bromoform, Chloromethane, Dibromomethane.
Which of the following compounds has the highest boiling point?
How many structural isomers are possible for a compound with the molecular formula C3H7Cl?
The decreasing order of boiling points of alkyl halides is:
Which of the following possesses the highest melting point?
Arrange the following compounds in the increasing order of their densities.
(a)
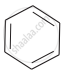
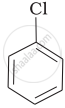
(b)
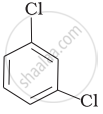
(c)
(d)
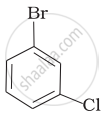
Which is the correct increasing order of boiling points of the following compounds?
1-Iodobutane, 1-Bromobutane, 1-Chlorobutane, Butane
Which is the correct increasing order of boiling points of the following compounds?
1-Bromoethane, 1-Bromopropane, 1-Bromobutane, Bromobenzene
Why alkyl halides though polar are immiscible with water?
Write the structure of the following organic halogen compound:
4-tert-Butyl-3-iodoheptane
