Advertisements
Advertisements
प्रश्न
Balance the equation stepwise.
NaOH(aq) + H2SO4(aq) → Na2SO4(aq) + H2O(l)
उत्तर
NaOH(aq) + H2SO4(aq) → Na2SO4(aq) + H2O(l)
Step1: Count the number of each atom in reactant side:
Na = 1
H = 3
O = 5
S = 1
Step 2: Count the number of each atom in product side:
Na = 2
H = 2
O = 5
S = 1
Step3: Then balance the number of each atom in an equation by multiplying reactant and product side with numeral value:
If we multiply NaOH by 2 in the reactant side and H2O by 2 in the product side, then number of atoms in product and reactant side gets balance.
2NaOH(aq) + H2SO4(aq) → Na2SO4(aq) +2H2O(l)
APPEARS IN
संबंधित प्रश्न
On what basis is a chemical equation balanced?
Complete and balance the following equation:
CA (OH)2 + .............  CaCO3 + H2O
CaCO3 + H2O
Balance the given chemical equation:
C + CO2 → CO
What are the various ways in which a chemical equation can be made more informative? Give examples to illustrate your answer.
Rewrite the following information in the form of a balanced chemical equation:
Magnesium burns in carbon dioxide to form magnesium oxide and carbon.
What do you understand by exothermic reactions?
Is Electrolysis of water an endothermic reaction or an exothermic reaction?
Which of the following does not involve a chemical reaction?
When a mixture of gases X and Y is compressed to 300 atm pressure and then passed over a catalyst consisting of a mixture of zinc oxide and chromium oxide (heated to a temperature of 300°C), then an organic compounds Z having the molecular formula CH4O is formed. X is a highly poisonous gas which is formed in appreciable amounts when a fuel burns in a limited supply of air; Y is a gas which can be made by the action of a dilute acid on an active metal; and Z is a liquid organic compound which can react with sodium metal to produce hydrogen gas.
(a) What are X, Y and Z?
(b) Write a balanced chemical equation of the reaction which takes place when X and Y combine to form Z. Indicate the conditions under which the reaction occurs.
A white precipitate will be formed if we add common salt solution to:
(a) Ba(NO3)2 solution
(b) KNO3 solution
(c) AgNO3 solution
(d) Mg(NO3)2 solution
Explain the following reaction with one suitable example .
Decomposition reaction
Balance the following chemical equation :
FeS + HCl → FeCl2 + H2S
Balance the following chemical equation :
H2 + O2 → H2O
Write your observation for the following chemical reaction and name the product formed :
When dilute acetic acid is poured on baking soda.
Write symbolic representation for the following word equation and balance them :
Aluminium + Chlorine → Aluminium chloride.
Dry raisins were soaked in water for 2 hours, to determine the percentage of water absorbed by raisins. Before final weighing of swollen raisins, the extra water left on the surface of soaked raisins was removed by:
Write the chemical equation for the following word equation and balance them.
Magnesium + Sulphuric acid → Magnesium sulphate + Hydrogen
Balance the following equation:
Zn + KOH → K2ZnO2 + H2
Balance the following equation:
MnO2 + HCl → MnCl2 + H2O + Cl2
Balance the following equation:
Pb3O4 + HCl → PbCl2 + H2O + Cl2
Write the balanced chemical equation of the following reaction.
zinc sulphide + oxygen → zinc oxide + sulphur dioxide
Write the balanced chemical equation of the following reaction.
aluminium sulphate + sodium hydroxide → sodium sulphate + sodium meta aluminate + water.
\[\ce{MnO2 + 4HCl -> MnCl2 + 2H2O + Cl2}\]
0.02 moles of pure MnO2 is heated strongly with conc. HCl. Calculate mass of MnO2 used.
Write word equation for the following molecular equation:
FeSO4 + 2NaOH →Na2SO4 + Fe(OH)2 ↓
Word equation:
State why [↓] indication is seen after Fe(OH)2 and not after Na2SO4
Write word equation for the following molecular equation:
CaO + H20 → Ca (OH)2 + Δ
Word equation:
State why [+ Δ ] is not written in the reaction, if ZnO reacts with H2O.
A chemical reaction is generally accompanied by certain external indications or characteristics. These include – change of – (a) colour (b) state (c) smell (d) evolution of gas (e) formation of precipitate (f) evolution or absorption of heat. With reference to change of colour – state the change in colour seen when the following are heated – lead [IV] oxide.
In certain reaction a change of state is observed i.e. solid to liquid, liquid to gas etc. – State the change of state of the products – to give the respective reactant.
\[\ce{2H2O ->[electric][current] 2H2 + O2}\]
In certain reaction an insoluble solid called precipitate is formed. State the colour and name of the precipitate formed in the following reaction involving addition of:
Iron [II] sulphate to sodium hydroxide.
Give one example in the case where supplying energy [given below] is necessary for a chemical reaction.
Pressure
Name the following:
Two gases which react under pressure in presence of a catalyst at elevated temperatures to give a gaseous product.
Give reason for the following:
Silver salts are kept in dark coloured bottles.
Write word equation for the following chemical reaction given below. Also state the observation seen in the case.
Pb(NO3)2 + 2NH4OH → 2NH4N03 + Pb(OH)2↓
Write word equation for the following chemical reaction given below. Also state the observation seen in the case.
\[\ce{2SO2 + O2 ⇌[V2O5][450°C] 2SO3}\]
An element X is trivalent. Write the balanced equation for the combustion of X in oxygen.
Balance the following simple equation:
ZnS + O2 → ZnO + SO2
Balance the following simple equation:
NaOH + Cl2 → NaCl + NaClO + H2O
Write a balanced equation for the following word equation:
Nitrogen dioxide + Water + Oxygen → Nitric acid
The chemical equation Na2SO4(aq) + BaCl2(aq) → BaSO4(s)↓ + 2NaCl(aq) represents which of the following types of reaction?
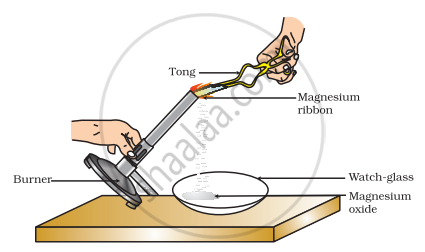
Which of the following is the correct observation of the reaction shown in the above set up?
