Advertisements
Advertisements
प्रश्न
Balance the following ionic equations.
\[\ce{MnO^{-}4 + H^{+} + Br^{-} -> Mn^{2+} + Br2 + H2O}\]
उत्तर
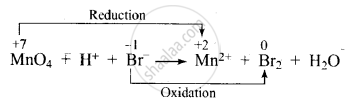
Dividing the equation into two half-reactions:
Oxidation half-reaction: \[\ce{Br^{-} -> Br2}\]
Reduction half-reaction: \[\ce{MnO^{-}4 -> Mn^{2+}}\]
Balancing oxidation and reduction half-reactions separately as:
Oxidation half-reaction:
\[\ce{Br^{-} -> Br2}\]
\[\ce{2Br^{-} -> Br2}\]
\[\ce{2Br^{-} -> Br2 + 2e-}\] .....(i)
Reduction half-reaction:
\[\ce{MnO^{-}4 -> Mn^{2+}}\]
\[\ce{MnO^{-}4 + 5e^{-} -> Mn^{2+}}\]
\[\ce{MnO^{-}4 + 8H^{+} + 5e^{-} -> Mn^{2+}}\]
\[\ce{MnO^{-}4 + 8H^{+} + 5e^{-} -> Mn^{2+} + 4H2O}\] .....(ii)
To balance the electrons, multiply equation (i) by 5 and equation (ii) by 2 and add
\[\ce{2MnO^{-}4 + 10Br^{-} + 16H^{+} -> 2Mn^{2+} + 5Br2 + 8H2O}\]
APPEARS IN
संबंधित प्रश्न
Balance the following equation in basic medium by ion-electron method and oxidation number methods and identify the oxidising agent and the reducing agent.
\[\ce{P4(s) + OH–(aq) —> PH3(g) + HPO^–_2(aq)}\]
Balance the following equation in the basic medium by ion-electron method and oxidation number methods and identify the oxidising agent and the reducing agent.
\[\ce{N2H4(l) + ClO^-_3 (aq) → NO(g) + Cl–(g)}\]
Balance the following reaction by oxidation number method.
\[\ce{H2SO4_{(aq)} + C_{(s)}->CO2_{(g)} + SO2_{(g)} + H2O_{(l)}(acidic)}\]
Balance the following equations by the oxidation number method.
\[\ce{I2 + NO^{-}3 -> NO2 + IO^{-}3}\]
Identify the redox reactions out of the following reactions and identify the oxidising and reducing agents in them.
\[\ce{HgCl2 (aq) + 2KI (aq) -> HgI2 (s) + 2KCl (aq)}\]
Identify the redox reactions out of the following reactions and identify the oxidising and reducing agents in them.
\[\ce{Fe2O3 (s) + 3CO (g) ->[Δ] 2Fe (s) + 3CO2 (g)}\]
Balance the following ionic equations.
\[\ce{MnO^{-}4 + SO^{2-}3 + H^{+} -> Mn^{2+} + SO^{2-}4 + H2O}\]
In \[\ce{Cu^{2+} + Ag -> Cu + Ag^+}\], oxidation half-reaction is:
The weight of CO is required to form Re2(CO)10 will be ______ g, from 2.50 g of Re2O7 according to given reaction
\[\ce{Re2O7 + CO -> Re2(CO)10 + CO2}\]
Atomic weight of Re = 186.2; C = 12 and O = 16.
Consider the following reaction:
\[\ce{xMnO^-_4 + yC2O^{2-}_4 + zH^+ -> xMn^{2+} + 2{y}CO2 + z/2H2O}\]
The values of x, y, and z in the reaction are, respectively:
