Advertisements
Advertisements
प्रश्न
Balance the following equations by the oxidation number method.
\[\ce{I2 + NO^{-}3 -> NO2 + IO^{-}3}\]
उत्तर
We can balance the given equation by oxidation number method-
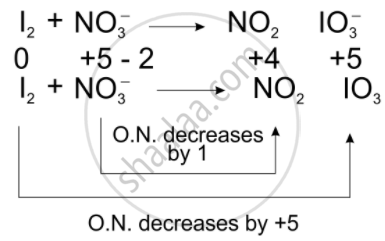
Total decrease in O.N. = 1
To equilize O.N. multiply \[\ce{NO^{-}3}\], by 10
\[\ce{I2 + 10 NO^{-}3 -> 10NO2 + IO^{-}3}\]
Balancing atoms other than \[\ce{O}\] and \[\ce{H}\]
\[\ce{I2 + 10NO^{-}3 -> 10NO2 + 2IO^{-}3}\]
Balancing \[\ce{O}\] and \[\ce{H}\]
\[\ce{I2 + 10NO^{-}3 + 8H+ -> 10NO2 + 2IO^{-}3 + 4H2O}\]
APPEARS IN
संबंधित प्रश्न
The compound AgF2 is an unstable compound. However, if formed, the compound acts as a very strong oxidizing agent. Why?
Balance the following redox reactions by ion-electron method:
- \[\ce{MnO-_4 (aq) + I– (aq) → MnO2 (s) + I2(s) (in basic medium)}\]
- \[\ce{MnO-_4 (aq) + SO2 (g) → Mn^{2+} (aq) + HSO-_4 (aq) (in acidic solution)}\]
- \[\ce{H2O2 (aq) + Fe^{2+} (aq) → Fe^{3+} (aq) + H2O (l) (in acidic solution)}\]
- \[\ce{Cr_2O^{2-}_7 + SO2(g) → Cr^{3+} (aq) + SO^{2-}_4 (aq) (in acidic solution)}\]
Balance the following reaction by oxidation number method.
\[\ce{MnO^-_{4(aq)} + Br^-_{ (aq)}->MnO2_{ (s)} + BrO^-_{3(aq)}(basic)}\]
Identify the oxidising agent in the following reaction:
\[\ce{CH4_{(g)} + 2O2_{(g)} -> CO2_{(g)} + 2H2O_{(l)}}\]
Identify the redox reactions out of the following reactions and identify the oxidising and reducing agents in them.
\[\ce{PCl3 (l) + 3H2O (l) -> 3HCl (aq) + H3PO3 (aq)}\]
Balance the following ionic equations.
\[\ce{MnO^{-}4 + SO^{2-}3 + H^{+} -> Mn^{2+} + SO^{2-}4 + H2O}\]
Balance the following ionic equations.
\[\ce{MnO^{-}4 + H^{+} + Br^{-} -> Mn^{2+} + Br2 + H2O}\]
The weight of CO is required to form Re2(CO)10 will be ______ g, from 2.50 g of Re2O7 according to given reaction
\[\ce{Re2O7 + CO -> Re2(CO)10 + CO2}\]
Atomic weight of Re = 186.2; C = 12 and O = 16.
Consider the following reaction:
\[\ce{xMnO^-_4 + yC2O^{2-}_4 + zH^+ -> xMn^{2+} + 2{y}CO2 + z/2H2O}\]
The values of x, y, and z in the reaction are, respectively:
\[\ce{H2O2 -> 2H^+ + O2 + 2e^-}\]; E0 = −0.68 V.
This equation represents which of the following behaviour of H2O2?
