Advertisements
Advertisements
प्रश्न
Calculate mass defect and binding energy per nucleon of `"_10^20 Ne`, given
Mass of `"_10^20 Ne= 19.992397` u
Mass of `"_0^1H = 1.007825` u
Mass of `"_0^1n = 1.008665` u
उत्तर
Mass defect (Δm) = Mass of nucleons − Mass of nucleus
= (10 × 1.007825 + 10 × 1.008665)u - 19.992397u
= 0.172503u
Binding energy ΔE = 0.1725034 × 931 MeV
= 160.600 MeV
Binding energy per nucleon `(ΔE)/A = 160.600/20`
= 8.03 MeV
संबंधित प्रश्न
Is the nucleus formed in the decay of the nucleus `""_11^22Na`, an isotope or isobar?
Obtain the binding energy of the nuclei `""_26^56"Fe"` and `""_83^209"Bi"` in units of MeV from the following data:
`"m"(""_26^56"Fe")` = 55.934939 u
`"m"(""_83^209"Bi")`= 208.980388 u
Consider the fission of `""_92^238"U"` by fast neutrons. In one fission event, no neutrons are emitted and the final end products, after the beta decay of the primary fragments, are `""_58^140"Ce"` and `""_44^99"Ru"`. Calculate Q for this fission process. The relevant atomic and particle masses are
`"m"(""_92^238"U")` = 238.05079 u
`"m"(""_58^140"Ce")` = 139.90543 u
`"m"(""_44^99"Ru")` = 98.90594 u
What characteristic property of nuclear force explains the constancy of binding energy per nucleon (BE/A) in the range of mass number ‘A’ lying 30 < A < 170?
In a nuclear reactor, what is the function of:
(i) The moderator
(ii) The control rods
(iii) The coolant
The figure shows the plot of binding energy (BE) per nucleon as a function of mass number A. The letters A, B, C, D, and E represent the positions of typical nuclei on the curve. Point out, giving reasons, the two processes (in terms of A, B, C, D, and E ), one of which can occur due to nuclear fission and the other due to nuclear fusion.
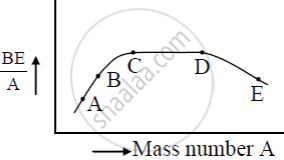
Answer the following question.
Draw the curve showing the variation of binding energy per nucleon with the mass number of nuclei. Using it explains the fusion of nuclei lying on the ascending part and fission of nuclei lying on the descending part of this curve.
A body's centre of mass
He23 and He13 nuclei have the same mass number. Do they have the same binding energy?
Find the binding energy of a H-atom in the state n = 2
