Advertisements
Advertisements
प्रश्न
Choose the correct alternative and rewrite the following sentence.
Which of the following is not required to find the pH of a given solution?
पर्याय
pH paper
Litmus paper
Standard pH value chart
- Universal indicator
उत्तर
Litmus paper
APPEARS IN
संबंधित प्रश्न
Translate the following statement into chemical equation and then balance it.
Potassium metal reacts with water to give potassium hydroxide and hydrogen gas.
Write the balanced chemical equation for the following reaction.
\[\ce{Barium chloride + Potassium sulphate → Barium sulphate + Potassium chloride}\]
2 mL of sodium hydroxide solution is added to a few pieces of granulated zinc metal taken in a test tube. When the contents are warmed, a gas evolves which is bubbled through a soap solution before testing. Write the equation of the chemical reaction involved and the test to detect the gas. Name the gas which will be evolved when the same metal reacts with dilute solution of a strong acid.
State whether the following statement is true or false:
A chemical equation can be balanced easily by altering the formula of a reactant or product.
How will you indicate Exothermic reaction in a chemical equation?
When hydrogen is passed over copper oxide, copper and steam are formed. Write a balanced equation for this reaction and state which of the chemicals are compounds.
When hydrogen is passed over copper oxide, copper and steam are formed. Write a balanced equation for this reaction and state which of the chemicals are metals.
Write balanced chemical equation from the following information:
An aqueous calcium hydroxide solution (lime water) reacts with carbon dioxide gas to produce a solid calcium carbonate precipitate and water.
Explain, with example, how the physical states of the reactants and products can be shown in a chemical equation.
Balance the following equation and add state symbols:
Zn + HCI → ZnCI2 + H2
What do you understand by endothermic reactions?
Balance the following chemical equation:
Write your observations and name the product when
Iron nails are added to an aqueous solution of copper sulphate.
Write symbolic representation for the following word equation and balance them :
Iron + Sulphur → Iron sulphide.
Balance the following equation :
Fe + O2 → Fe3 O4
Balance the following equation .
N2 + O2 → NO
Complete the following equation:
CH2 + H5OH `("Hot Conc." H_2SO_4)/`>
Complete the following equation:
CH3COOH + NaOH →
Balance the equation stepwise.
H2S2O7(l) + H2O(l) → H2SO4(l)
Write the chemical equation for the following word equation and balance them.
Nitrogen + Oxygen → Nitrogen monoxide
Balance the following equation:
NO2 +H2O → HNO2 + HNO3
What is the valency of :
carbon in CH4
Write the name and molecular formula of a carbon compound having its name suffixed with "-ol" and having two carbon atoms in its molecule. With the help of a chemical equation indicate what happens when this compound is heated with excess conc. H2SO4.
Write word equation for the following molecular equation:
\[\ce{N2 + 3H2 ⇌[Fe - 450°C][200 atoms] 2NH3 + \triangle}\]
Word equation:
State why [+ Δ] indication is seen after NH3.
State what 200 atmospheres – indicates.
State the function of Fe in the above reaction.
Give a balanced equation for addition of iron to copper [II] sulphate solution. State the change in colour seen.
Give an example of a chemical equation in which two reactants form:
one product
Balance the following simple equation:
ZnS + O2 → ZnO + SO2
Balance the following simple equation:
Fe2O3 + CO → Fe + CO2
Balance the following simple equation:
CaO + HCl → CaCl2 + H2O
Write a balanced equation for the following word equation:
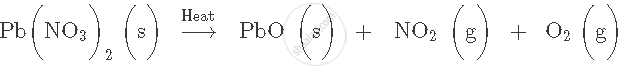
Lead dioxide [lead (IV) oxide] → Lead monoxide + Oxygen
Write a balanced equation for the following word equation:
Aluminium + Oxygen → Aluminium oxide
Write a balanced equation for the following word equation:
Potassium bicarbonate → Potassium carbonate + Water + Carbon dioxide
Underline the compound in the equation given below, it is incorrectly balanced and write the correct balancing for the same.
CaC2 + N2 → 2CaCN2 + C
Balance the given chemical reaction as per the instructions below.
\[\ce{NaOH + H2SO4 -> Na2SO4 + H2O}\]
Make a list of the elements in the chemical equation.
Which of the following is not a physical change?
Which of the following statements about the given reaction are correct?
`3"Fe"("s") + 4"H"_2"O"("g") -> "Fe"_3"O"_4("s") + 4"H"_2("g")`
- Iron metal is getting oxidised
- Water is getting reduced
- Water is acting as reducing agent
- Water is acting as oxidising agent
