Advertisements
Advertisements
प्रश्न
Balance the equation stepwise.
H2S2O7(l) + H2O(l) → H2SO4(l)
उत्तर
H2S2O7(l) + H2O(l) → H2SO4(l)
Step 1: Count the number of each atom on the reactant side:
| Elements | No. of atoms in the reactant |
| H | 4 |
| S | 2 |
| O | 8 |
Step 2: Count the number of each atom on the product side:
| Elements | No. of atoms in the product |
| H | 2 |
| S | 1 |
| O | 4 |
Step 3: Number of atoms of elements on both side is not the same.
If we multiply the product side by 2, then the number of atoms in the product and reactant side gets balanced.
| Elements | No. of atoms in the reactant | No. of atoms in the product |
| H | 4 | 2 × 2 = 4 |
| S | 2 | 1 × 2 = 2 |
| O | 8 | 4 × 2 = 8 |
H2S2O7(l) + H2O(l) → 2H2SO4(l)
APPEARS IN
संबंधित प्रश्न
Balance the following chemical equation.
\[\ce{NaOH + H2SO4 -> Na2SO4 + H2O}\]
What does the symbol (aq) represent in a chemical equation?
Fill in the following blank with suitable word:
A solution made in water is known as an ........... solution and indicated by the symbol ...........
When hydrogen is passed over copper oxide, copper and steam are formed. Write a balanced equation for this reaction and state which of the chemicals are metals.
When hydrogen is passed over copper oxide, copper and steam are formed. Write a balanced equation for this reaction and state which of the chemicals are non-metals.
What are the various ways in which a chemical equation can be made more informative? Give examples to illustrate your answer.
State one characteristic of the chemical reaction which takes place when lemon juice is added gradually to potassium permanganate solution.
State one characteristic of the chemical reaction which takes place when quicklime is treated with water
Which of the following is not an endothermic reaction?
(a) CaCO3 → CaO + CO2
(b) 2H2O →2H2 + O2
(c) 6CO2 + 6H2O → C6H12O6 + 6O2
(d) C6H12O6 + 6O2 → 6CO2 + 6H2O
Name the various types of chemical reactions.
Write word equation for the following skeletal equation:
\[\ce{CO + O2 -> CO2}\]
Balance the following chemical equation :
FeS + HCl → FeCl2 + H2S
Balance the following chemical equation :
Na20 + H20 → NaOH
Balance the following equation. Also name the product formed.
`"Na"+ "H"_2 "O" → "NaOH"+ "H"_2`
Balance the following equation. Also name the product formed.
N2 + H2 → NH3
Dry raisins were soaked in water for 2 hours, to determine the percentage of water absorbed by raisins. Before final weighing of swollen raisins, the extra water left on the surface of soaked raisins was removed by:
Raisins are wiped off gently before final weighing with the help of
Balance the equation stepwise.
NaOH(aq) + H2SO4(aq) → Na2SO4(aq) + H2O(l)
Balance the following equation:
PbS + O2 → PbO + SO2
Balance the following equation:
MnO2 + HCl → MnCl2 + H2O + Cl2
Balance the following equation:
H2O + Cl2 → HCl + O2
Write the balanced chemical equation of the following reaction.
barium chloride + sulphuric acid → barium sulphate + hydrochloric acid
Write the balanced chemical equation of the following reaction.
zinc sulphide + oxygen → zinc oxide + sulphur dioxide
(a) Give only one suitable chemical test to identify the following gases.
- Ammonia
- Sulphur dioxide
- Hydrogen Chloride
- Chlorine
- Carbon Dioxide
- Oxygen
- Hydrogen
(b) Select a basic gas mentioned in Q.1 (a). How is the basic nature suspected?
(c)Select acidic gases from the gases mentioned in Q.1 (a). How is the acidic nature suspected?
(d) State the gas responsible for bleaching action.
(e)Which gas turn blue cobalt chloride paper light pink?
Considering the relationship in the first pair, complete the second paie :
AgCI : White :: CuI2 : ___________
Define: Endothermic reaction
Name the substances that are getting oxidised and reduced in the process.
When a potassium iodide solution is added to a solution of lead (II) nitrate in a test tube, a precipitate is formed.
Write the balanced chemical equation for this reaction.
Balance the following simple equation:
Mg + N2 → Mg3N2
Balance the following simple equation:
Fe + Cl2 → FeCl3
Balance the following simple equation:
H2S + Cl2 → S + HCl
Underline the compound in the equation given below, it is incorrectly balanced and write the correct balancing for the same.
Fe2O3 + 2H2 → 2Fe + 3H2O
Underline the compound in the equation given below, it is incorrectly balanced and write the correct balancing for the same.
6NaOH + 3Cl2 → 6NaCl + NaClO3 + 3H2O
Writing a chemical reaction in brief by using chemical formulae is called as _______.
The chemical equation Na2SO4(aq) + BaCl2(aq) → BaSO4(s)↓ + 2NaCl(aq) represents which of the following types of reaction?
Three beakers labelled as A, B and C each containing 25 mL of water were taken. A small amount of NaOH, anhydrous CuSO4 and NaCl were added to the beakers A, B and C respectively. It was observed that there was an increase in the temperature of the solutions contained in beakers A and B, whereas in case of beaker C, the temperature of the solution falls. Which one of the following statement(s) is(are) correct?
- In beakers A and B, exothermic process has occurred.
- In beakers A and B, endothermic process has occurred.
- In beaker C exothermic process has occurred.
- In beaker C endothermic process has occurred.
What is a product in a chemical equation?
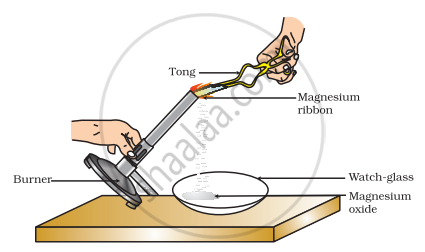
Which of the following is the correct observation of the reaction shown in the above set up?
Complete the following blank in the equation as indicated.
\[\ce{CaH2_{(s)} + 2H2O_{( aq)}-> Ca(OH)2_{(s)} + 2H2_{(g)}}\]
Grams: 42 g + ______ `→` ______ + ______
