Advertisements
Advertisements
Question
Balance the equation stepwise.
H2S2O7(l) + H2O(l) → H2SO4(l)
Solution
H2S2O7(l) + H2O(l) → H2SO4(l)
Step 1: Count the number of each atom on the reactant side:
| Elements | No. of atoms in the reactant |
| H | 4 |
| S | 2 |
| O | 8 |
Step 2: Count the number of each atom on the product side:
| Elements | No. of atoms in the product |
| H | 2 |
| S | 1 |
| O | 4 |
Step 3: Number of atoms of elements on both side is not the same.
If we multiply the product side by 2, then the number of atoms in the product and reactant side gets balanced.
| Elements | No. of atoms in the reactant | No. of atoms in the product |
| H | 4 | 2 × 2 = 4 |
| S | 2 | 1 × 2 = 2 |
| O | 8 | 4 × 2 = 8 |
H2S2O7(l) + H2O(l) → 2H2SO4(l)
APPEARS IN
RELATED QUESTIONS
Why should a magnesium ribbon be cleaned before it is burnt in air?
Correct and balance the following equation:
N + H  NH3
NH3
Write complete balanced equation for the following reaction:
Calcium (solid) + water (liquid)  Calcium Hydroxide (solution) + Hydrogen (gas)
Calcium Hydroxide (solution) + Hydrogen (gas)
Balance the given equation:
NH3 + CuO  Cu +N2 +H2O
Cu +N2 +H2O
When hydrogen is passed over copper oxide, copper and steam are formed. Write a balanced equation for this reaction and state which of the chemicals are compounds.
Give one example of a chemical reaction characterised by formation of a precipitate.
Give one example of an exothermic reaction.
The chemical reaction between quicklime and water is characterised by:
(a) evolution of hydrogen gas
(b) formation of slaked lime precipitate
(c) change in temperature of mixture
(d) change in colour of the product
When the solution of substance X is added to a solution of potassium iodide, then a yellow solid separates out from the solution.
(a) What do you think substance X is likely to be?
(b) Name the substance which the yellow solid consists of.
(c) Which characteristic of chemical reaction is illustrated by this example?
(d) Write a balanced chemical equation for the reaction which takes place. Mention the physical states of all the reactant and products involved in the chemical equation.
When metal X is treated with a dilute acid Y, then a gas Z is evolved which burns readily by making a little explosion.
(a) Name any two metals which can behave like metal X.
(b) Name any two acids which can behave like acid Y.
(c) Name the gas Z.
(d) Is the gas Z lighter than or heavier than air?
(e) Is the reaction between metal X and dilute acid Y, exothermic or endothermic?
(f) By taking a specific example of metal X and dilute acid Y, write a balanced chemical equation for the reaction which takes place. Also indicate physical states of all the reactants and products.
Balance the following chemical equation:
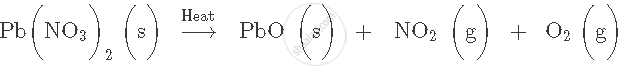
Name the type of chemical reaction shown by the following equation:
2Mg+ O2 → 2MgO
Write the balanced equation for the following chemical reaction.
\[\ce{Barium chloride + Aluminium sulphate -> Barium sulphate + Aluminium chloride}\]
Write word equation for the following skeletal equation:
\[\ce{Ca + O2 → CaO}\]
Write your observation and name the product when zinc reacts with dilute hydrochloric acid.
Dry raisins were soaked in water for 2 hours, to determine the percentage of water absorbed by raisins. Before final weighing of swollen raisins, the extra water left on the surface of soaked raisins was removed by:
Raisins are wiped off gently before final weighing with the help of
Write down what you understood from the following chemical reaction.
AgNO3(aq) + NaCI(aq) → AgCI ↓ + NaNO3(aq)
Write the answer to the following.
Explain the similarity and difference in two events, namely adding NaOH to water and adding CaO to water.
Write the chemical equation for the following word equation and balance them.
Calcium + Nitrogen → Calcium nitride
Balance the following equation:
Fe + H2O → Fe3O4 + H2
Balance the following equation:
NO2 +H2O → HNO2 + HNO3
Balance the following equation:
Zn + HNO3 `→` Zn(NO3)2 + H2O + NO2
Write the balanced chemical equation of the following reaction.
potassium bicarbonate + sulphuric acid → potassium sulphate + carbon dioxide + water
\[\ce{MnO2 + 4HCl -> MnCl2 + 2H2O + Cl2}\]
0.02 moles of pure MnO2 is heated strongly with conc. HCl. Calculate mass of MnO2 used.
In certain reaction a change of state is observed i.e. solid to liquid, liquid to gas etc. – State the change of state of the products – to give the respective reactant.
NH3 + HCl ⇌ NH4Cl
Representation of the results of a chemical change – is a chemical equation.
For the equation: FeCl3 + 3NH4OH 3NH4Cl + Fe(OH)3 ↓
Answer the following:
What is the indications of the arrow between the reactants and the products and of the arrow pointing downwards at the end.
Balance the following simple equation:
ZnS + O2 → ZnO + SO2
Balance the following simple equation:
H2 + Cl2 → HCl
Balance the following simple equation:
KHCO3 → K2CO3 + H2O + CO2
Balance the following simple equation:
\[\ce{N2 + H2 <=> NH3}\]
Fill in the blank with the correct word from the options given below:
A chemical equation is a shorthand form for a _______ change.
Underline the compound in the equation given below, it is incorrectly balanced and write the correct balancing for the same.
Na2SO3 + HCl → 2NaCl + H2O + SO2
Underline the compound in the equation given below, it is incorrectly balanced and write the correct balancing for the same.
Fe2O3 + 2H2 → 2Fe + 3H2O
Underline the compound in the equation given below, it is incorrectly balanced and write the correct balancing for the same.
PbO2 +4HCl → PbCl2 + H2O + Cl2
Complete the given chemical reaction.
\[\ce{CuSO_{4(aq)} + Fe_{(s)} ->}\]______ + ______
Write an Explanation.
Balanced equations
Balance the following chemical equation.
- Na + O2 → Na2O3
- Ca + N2 → Ca3N2
- N2 + H2 →NH3
- CaCO3 +HCl → CaCl2 + CO2 +H2O
- Pb(NO3)2 → PbO + NO2 + O2
What is a reactant in a chemical equation?
