Advertisements
Advertisements
प्रश्न
CO2 and H2O both are triatomic molecule but their dipole moment values are different. Why?
उत्तर
- A linear form of carbon dioxide has zero dipole moment. In CO2 the dipole moment of two polar bonds are equal in magnitude but have opposite directions. Hence, the net dipole moment of the CO2 molecule is
µ = µ1 + µ2
µ = µ1 + (- µ1) = 0
In this case `mu = vec mu_1 + vec((- mu_1) = 0` - But in the case of water, net dipole moment is the vector sum µ1 + µ2 as follows:
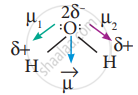
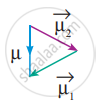
Dipole moment in water is found to be 1.85 D.
- CO2 and H2O both are triatomic molecules but their dipole moment values are zero and 1.85 D respectively.
APPEARS IN
संबंधित प्रश्न
Non – Zero dipole moment is shown by ______.
Define bond order.
What is dipole moment?
Linear form of carbondioxide molecule has two polar bonds. Yet the molecule has Zero dipole moment. Why?
Define bond energy.
Explain resonance with reference to a carbonate ion.
Explain the bond formation in ethylene.
Which one of the following has the highest bond order?
- N2
- N2+
- N2–
The bond order in NO is 2.5 while that in NO+ is 3. Which of the following statements is true for these two species?
Bond distance in HF is 9.17 × 10−11 m. Dipole moment of HF is 6.104 × 10−30 Cm. The percentage of ionic character in HF will be ______.
(electron charge = 1.60 × 10−19 C)
