Advertisements
Advertisements
प्रश्न
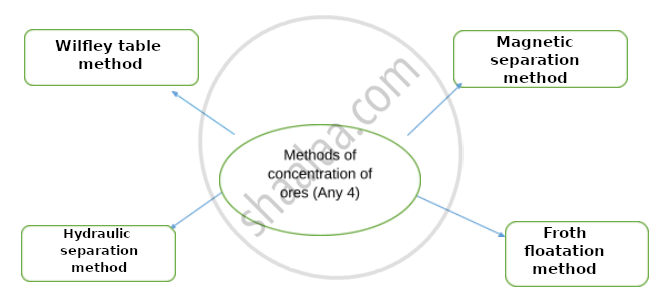
Complete flow chart given below.
उत्तर
APPEARS IN
संबंधित प्रश्न
Give the importance of the following for living beings:
Nitrogen
Metal A has an electronic configuration of 2, 8, 1 and metal B has 2, 8, 8, 2 which is more reactive metal.
Give the effect of heat on their: hydroxide
A student has been collecting silver coins and copper coins. One day she observed a black
coating on silver coins and a green coating on copper coins. Which chemical phenomenon is
responsible for these coatings? Write the names of black and green coatings.
An ore on being heated in air forms sulphurous anhydride. Write the process used for the concentration of this ore.
`HgS + O_2`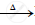 ..................
..................
Give the ionization reactions of electrolyte used in Hall’s process. write the reaction at the cathode and the anode. Why the anode has to be replaced in this process?
Where are the cathode and anode in the electrolytic cell? Name the material used for these?
Which metal is used for:
making pipes, buckets, water tanks,
Aluminium is extracted from its chief ore, bauxite. The ore is first purified and then the metal is extracted from it by electrolytic reduction.
Write three balanced equations for the purification of bauxite.
Explain the term of Metallurgy .
Name the following:
Name an allotrope of a non-metal that allows electricity to pass through it.
The following is a sketch of an electrolytic cell used in the extraction of aluminium :
(a) What is the substance of which the electrode A and B are made?
(b) At which electrode (A or B) is aluminium formed?
(c) What are the two aluminium compounds in the electrolyte C?
(d) Why is it necessary for electrode B to be continuously replaced?
The chemical formula of zinc blend is _______.
Write the molecular formulae of the following compound.
Stannic oxide
Explain concept with example/explain with the help of a balanced equation.
Gangue
Explain concept with example/explain with the help of a balanced equation.
Minerals
Observe the figure and name and explain in brief the following method:

State the reason for the addition of caustic alkali to bauxite ore during the purification of bauxite.
In leaching of alumina from bauxite by Bayer's process, then ore is treated with ______.
Calcination is used in metallurgy for the removal of?
