Advertisements
Advertisements
प्रश्न
Complete the following calculations. Show working for complete credit :
If the empirical formula of a compound is CH and it has a vapour density of 13, find the molecular formula of the compound.
उत्तर
For acetylene , molecular mass = 2 X V.D. = 2 x 13 = 26g
The empirical mass = 12 (c) + 1(H) = 13g
n = `"Molecular formula mass"/"Empirical formula weight"`
= `26/13` = 2
Molecular formula of acetylene = 2 x Empirical formula =C2H2
Similarly, for benzene molecular mass = 2 X V.D = 2 x 39 = 78
n = 78/13 = 6
So, the molecular formula = C6H6.
APPEARS IN
संबंधित प्रश्न
Give balanced chemical equations for the following conversions A, B, and C:
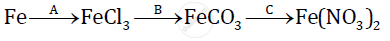
Prove the Following :
2 X V.D. = Molecular mass.
Calculate the percentage composition of oxygen in lead nitrate [Pb(NO3)2]. [Pb = 207, N= 14, O = 16]
What weight of sulphuric acid will be required to dissolve 3g of magnesium carbonate?
[Mg = 24, C =12, 0 = 16 ]
MgCO3 + H2SO4 → MgSO4 + H2O+ CO2
10g of NaCl solution is mixed with 17g of silver nitrate solution. Calculate the weight of silver chloride precipitated.
AgNO3 + NaCl → AgCI + NaNO3
Water can split into hydrogen and oxygen under suitable conditions. The equations representing the change is: 2H2O(I) → 2H2 (g) + O2(g)
Ammonia burns in oxygen and the combustion in the presence of a catalyst may be represented as:
2NH3 (g) +21/2O2 (g) → 2NO (g) + 3H2O (I)
What mass of steam is produced when 1.5 g of nitrogen monoxide is formed?
Calculate the percentage of nitrogen in aluminium nitride. [Al = 27, N = 14]
Which of the following contains maximum number of molecules?
Correct the statement, if required.
Under similar conditions of temperature and pressure, two volumes of hydrogen combined with two volumes of oxygen will give two volumes of water vapour.
A gas cylinder filled with hydrogen holds 5 g of the gas. The same cylinder holds 85 g of gas X under the same temperature and pressure. Calculate the vapour density of gas X.
