Advertisements
Advertisements
प्रश्न
Water can split into hydrogen and oxygen under suitable conditions. The equations representing the change is: 2H2O(I) → 2H2 (g) + O2(g)
Ammonia burns in oxygen and the combustion in the presence of a catalyst may be represented as:
2NH3 (g) +21/2O2 (g) → 2NO (g) + 3H2O (I)
What mass of steam is produced when 1.5 g of nitrogen monoxide is formed?
उत्तर
Given equation is:
2NH3 (g) +21/2O2 (g) → 2NO (g) + 3H2O (I)
Molecular mass of NO = 30
Molecular mass of H2O = 18
From the equation :
2 moles of NO = 3 moles of H2O
60g of NO = 54g of H2O.
1.5g of NO = 54 x 1.5 / 60 = 1.35g of H2O.
APPEARS IN
संबंधित प्रश्न
Give balanced chemical equations for the following conversions A, B, and C:
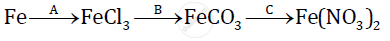
Aluminium carbide reacts with water according to the following equation :
`Al_4C_3 + 12H_2O-> 4Al(OH)_3 + 3CH_4`
1)What mass of aluminium hydroxide is formed from 12g of aluminium carbide?
2) What volume of methane at s.t.p. is obtained from 12g of aluminium carbide?
[Relatively molecular weight of `Al_4Cl_3 = 144; Al(OH)_3 = 78]`
Define the term: Vapour density.
Empirical formula of a compound is CH2O. If its empirical formula is equal to its vapour density, calculate the molecular formula of the compound.
Potassium nitrate on strong heating decomposes as under :
2KNO3 → 2KNO2 + O2
Calculate : Weight of potassium nitrite formed.
(K = 39, 0 = 16, N = 14)
Find the total percentage of oxygen in magnesium nitrate crystal Mg(NO3)2.6H2O.
[H = 1, N = 14, O = 16, Mg = 24]
Calculate the percentage of nitrogen in aluminium nitride. [Al = 27, N = 14]
A certain gas 'X' occupies a volume of 100 cm3 at S.T.P. and weighs 0.5 g. Find its relative molecular mass.
A gaseous hydrocarbon contains 82.76% of carbon. Given that its vapour density is 29, find its molecular formula.
[C = 12, H = 1]
Calculate the relative molecular mass of:
Potassium chlorate
