Advertisements
Advertisements
प्रश्न
Consider structures I to VII and answer the question:
| I. | CH3 – CH2 – CH2 – CH2 – OH |
| II. | |
| III. | |
| IV. | |
| V. | CH3 – CH2 – O – CH2 – CH3 |
| VI. | CH3 – O – CH2 – CH2 – CH3 |
| VII. |
Identify the pairs of compounds that represents position isomerism.
उत्तर
If two or more compounds differ in the position of substituent, functional group, or multiple bonds but molecular formula is same then these are said to be position isomers as they exhibit position isomerism.
In the given structures, I and II, III and IV, VI and VII are found to be the position isomers.
APPEARS IN
संबंधित प्रश्न
Write IUPAC name of the product obtained by the ozonolysis of the following compound:
2-Ethylbut-1-ene
What is the relationship between the members of following pairs of structures? Are they structural or geometrical isomers or resonance contributors?
Find out the type of isomerism exhibited by the following pair.
Find out the type of isomerism exhibited by the following pair.
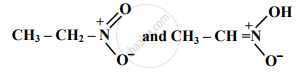
Molecular formula of the functional isomer of methyl formate is ____________.
But-1-ene and But-2-ene are examples of ____________.
Which of the following is a functional isomer of pentan-2-ol?
The type of isomerism possible in 2-butene is ____________.
What is the relationship between the members of following pairs of structures? Are they structural or geometrical isomers or resonance contributors?
In which of the following, functional group isomerism is not possible?
Which of the following pairs are not functional group isomers?
| I. | |
| II. | |
| III. | |
| IV. |
(i) II and III
(ii) II and IV
(iii) I and IV
(iv) I and II
Consider structures I to VII and answer the question:
| I. | CH3 – CH2 – CH2 – CH2 – OH |
| II. | |
| III. | |
| IV. | |
| V. | CH3 – CH2 – O – CH2 – CH3 |
| VI. | CH3 – O – CH2 – CH2 – CH3 |
| VII. |
Identify the pairs of compounds which are functional group isomers.
Assertion (A): Pent- 1- ene and pent- 2- ene are position isomers.
Reason (R): Position isomers differ in the position of functional group or a substituent.
Assertion (A): The compound cyclooctane has the following structural formula: ![]()
It is cyclic and has conjugated 8π-electron system but it is not an aromatic compound.
Reason (R): (4n + 2)π electrons rule does not hold good and ring is not planar.
Ether and alcohol are ______.
The compound which shows metamerism is ______
Acetamide is isomer of ______.
The correct stereochemical name of
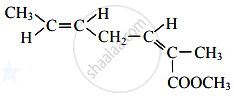
The number of acyclic structural isomers (including geometrical isomers) for pentene are ______.
Which of the following reactions will not produce a racemic product?
