Advertisements
Advertisements
प्रश्न
Correct the following statement :
Haematite is the chief ore of aluminium.
उत्तर
Bauxite is the chief ore of aluminium.
APPEARS IN
संबंधित प्रश्न
Write the equation for the reaction where the aluminum oxide for the electrolytic extraction of aluminum is obtained by heating aluminum hydroxide.
Name the constituents of Brass.
The reason for using Aluminium in the alloy duralumin is ______.
Why is food containing iron salts should not be cooked in aluminium utensils?
In order to obtain one tone of aluminium, the following inputs are required:
4 tones of bauxite, 150 Kg of sodium hydroxide and 600 Kg of graphite.
The aluminium compound in bauxite is aluminium oxide and the main impurity is iron (II) oxide. Aluminium is obtained by the electrolysis of aluminium oxide dissolved in cryolite.
In construction work, why is the alloy of aluminium, duralumin used rather than pure aluminium.
The sketch below illustrates the refin ing of aluminium by Hoope's process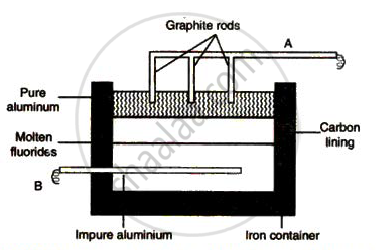
(a) Which of A and B is th e cathode and which one is the anode?
(b) What is the electroly te in the tank?
( c) What material is used for th e cathode?
Name the compound added to pure alumina to lower the fusion temperature during the electrolytic reduction of alumina.
Explain with reason:
A neutral gas other than oxygen is formed at the anode during the electrolysis of fused alumina.
Name the alloy used for the following purpose.
Surgical instruments
Fused alumina is reduced to aluminium by electrolytic reduction, since ______.
