Advertisements
Advertisements
प्रश्न
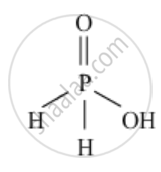
Draw the structures of `H_3PO_2`
उत्तर
`H_3PO_2`
APPEARS IN
संबंधित प्रश्न
Account for the following: Oxygen shows catenation behavior less than sulphur.
List the important sources of sulphur.
Why is H2O a liquid and H2S a gas?
Justify the placement of O, S, Se, Te and Po in the same group of the periodic table in terms of electronic configuration, oxidation state and hydride formation.
Why are halogens strong oxidising agents?
Explain why inspite of nearly the same electronegativity, oxygen forms hydrogen bonding while chlorine does not.
Match the items of Columns I and II and mark the correct option.
| Column I | Column II |
| (A) \[\ce{H2SO4}\] | (1) Highest electron gain enthalpy |
| (B) \[\ce{CCl3NO2}\] | (2) Chalcogen |
| (C) \[\ce{Cl2}\] | (3) Tear gas |
| (D) Sulphur | (4) Storage batteries |
Strong reducing behaviour of \[\ce{H3PO2}\] is due to ______.
These are physical properties of an elements.
- Sublimation enthalpy
- Ionisation enthalpy
- Hydration enthalpy
- Electron gain enthalpy
The total number of above properties that affect the reduction potential is ______. (Integer answer)
______ is a radioactive element in group 16 elements.
