Advertisements
Advertisements
प्रश्न
Draw the structures for Butanone
Draw the structures for the following compound.
Butanone
उत्तर
1)
\[\ce{CH3CH2COCH3}\]
2)
\begin{array}{cc}\\
\phantom{}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{O}\phantom{...}\ce{H}\phantom{}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{}|\phantom{....}|\phantom{}\\
\ce{H - C - C - C - C - H}\\
\phantom{}|\phantom{....}|\phantom{.........}|\phantom{}\\
\phantom{}\ce{H}\phantom{...}\ce{H}\phantom{........}\ce{H}\phantom{}
\end{array}
APPEARS IN
संबंधित प्रश्न
Write the name and general formula of a chain of hydrocarbons in which an addition reaction with hydrogen is possible. State the essential condition for an addition reaction. Stating this condition, write a chemical equation giving the name of the reactant and the product of the reaction.
Define the term ‘structural’ isomerism'.
List two reasons for carbon forming a large number of compounds. Name the type of bonding found in most of its compounds. Why does carbon form compounds mainly by this kind of bonding?
What is meant by isomers?
Draw the structures for Ethanoic acid.
Explain why propane cannot exhibit the structural isomerism property.
Fill in the following blank with suitable word:
Compounds of carbon with hydrogen alone are called ..........
Which of the following is the molecular formula of benzene?
C6H6, C6H10, C6H12, C6H14
What are hydrocarbons? Explain with examples.
How many isomers of the following hydrocharbons are possible?
C4H10
A diamond-toothed saw is usually used for cutting:
(a) steel girders
(b) logs of wood
(c) marble slabs
(d) asbestos sheets
The hydrocarbon which has alternate single and double bonds arranged in the form of a ring is:
(a) cyclobutane
(b) benzene
(c) butene
(d) hexene
Two organic compounds A and B have the same molecular formula C6H12. Write the names and structural formulae:
Which compound contains single bonds as well as a double bond?
The solid element A exhibits the property of catenation. It is also present in the form of a gas B in the air which is utilised by plants in photosynthesis. An allotrope C of this element is used in glass cutters.
(a) What is element A?
(b) What is the gas B?
(c) Name the allotrope C.
(d) State another use of allotrope C (other than in glass cutters).
(e) Name another allotrope of element A which exists as spherical molecules.
(f) Name a yet another allotrope of element A which conduct electricity.
A reagent which can help us to distinguish between alkenes and alkynes is ______.
The number of isomers of pentane is ______.
Draw two structural isomers of butane.
Name the following:
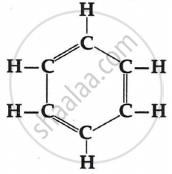
Draw the structure of propanone.
