Advertisements
Advertisements
प्रश्न
Explain in detail the kinetic interpretation of temperature.
उत्तर
To understand the microscopic origin of temperature in the same way, from pressure exerted by a gas,
P = `1/3 "N"/"V""m"bar("v"^2)`
PV = `1/3"Nm"bar("v"^2)` ............(1)
Comparing the equation (1) with the ideal gas equation PV = NkT,
NkT = `1/3"Nm"bar("v"^2)`
kT = `1/3"m"bar("v"^2)` .........(2)
Multiply the above equation by `3/2` on both sides,
`3/2"kT" = 1/2"m"bar("v"^2)` ...........(3)
R.H.S. of the equation (3) is called average kinetic energy of a single molecule `(bar"KE")`.
The average kinetic energy per molecule
`bar"KE"` = ∈ = `3/2"kT"` ...........(4)
Equation (3) implies that the temperature, of a gas, is a measure of the average translational kinetic energy per molecule of the gas.
Equation (4) is a very important result of the kinetic theory of gas. We can infer the following from this equation.
(i) The average kinetic energy of the molecule is directly proportional to the absolute temperature of the gas. Equation (3) gives the connection between the macroscopic world (temperature) to the microscopic world (motion of molecules).
(ii) The average kinetic energy of each molecule depends only on the temperature of the gas hot on the mass of the molecule. In other words, if the temperature of an ideal gas is measured using the thermometer, the average kinetic energy of each molecule can be calculated without seeing the molecule through the naked eye.
By multiplying the total number of gas molecules with the average kinetic energy of each molecule, the internal energy of the gas is obtained.
Internal energy of ideal gas U = `"N"(1/2"m"bar("v"^2))"`
By using equation (3), U = `3/2"NkT"` ..........(5)
Here, we understand that the internal energy of an ideal gas depends only on absolute temperature and is independent of pressure and volume.
APPEARS IN
संबंधित प्रश्न
The mean square speed of the molecules of a gas at absolute temperature T is proportional to
Find the number of molecules of an ideal gas in a volume of 1.000 cm3 at STP.
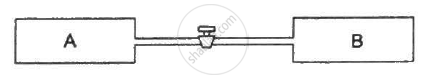
Figure shows two vessels A and B with rigid walls containing ideal gases. The pressure, temperature and the volume are pA, TA, V in the vessel A and pB, TB, V in the vessel B. The vessels are now connected through a small tube. Show that the pressure p and the temperature T satisfy `Ρ/T = 1/2 ({P_A}/{T_A}+{P_B}/{T_B))` when equilibrium is achieved.
Answer in brief:
Show that rms velocity of an oxygen molecule is `sqrt2` times that of a sulfur dioxide molecule at S.T.P.
When a gas is heated, its temperature increases. Explain this phenomenon on the basis of the kinetic theory of gases.
Calculate the average molecular kinetic energy
- per kmol
- per kg
- per molecule
of oxygen at 127°C, given that the molecular weight of oxygen is 32, R is 8.31 J mol−1K−1 and Avogadro’s number NA is 6.02 × 1023 molecules mol−1.
Calculate the value of λmax for radiation from a body having a surface temperature of 3000 K. (b = 2.897 x 10-3 m K)
If the density of nitrogen is 1.25 kg/m3 at a pressure of 105 Pa, find the root mean square velocity of nitrogen molecules.
When photons of energy hv fall on a metal plate of work function 'W0', photoelectrons of maximum kinetic energy 'K' are ejected. If the frequency of the radiation is doubled, the maximum kinetic energy of the ejected photoelectrons will be ______.
Average kinetic energy of H2 molecule at 300K is 'E'. At the same temperature, average kinetic energy of O2 molecule will be ______.
