Advertisements
Advertisements
प्रश्न
For bauxite, explain its significance in the extraction of aluminium.
उत्तर
- Aluminium is extracted from bauxite \[\ce{Al2O3·2H2O}\].
- Bauxite contains 60% \[\ce{Al2O3}\], the rest being sand, ferric oxide and titanium oxide.
APPEARS IN
संबंधित प्रश्न
Aluminium is said to be more reactive than iron, towards oxygen (or air) yet iron undergoes corrosion to a greater extent than aluminum. Explain.
Give reason for the following:
Nitric acid can be stored in aluminium containers.
How is aluminium obtained from pure ore?
In the Hall's process for extraction of aluminium, Give the formula and purpose of fluorspar and cryolite
Aluminium is a more active metal than iron, but suffers less corrosion. Why?
In order to obtain one tone of aluminium, the following inputs are required:
4 tones of bauxite, 150 Kg of sodium hydroxide and 600 Kg of graphite.
The aluminium compound in bauxite is aluminium oxide and the main impurity is iron (II) oxide. Aluminium is obtained by the electrolysis of aluminium oxide dissolved in cryolite.
In construction work, why is the alloy of aluminium, duralumin used rather than pure aluminium.
Answer the following question :
Name a metal which is found abundantly in earth's crust.
Describe the role played in the extraction of aluminum:
Cryolite
Write the balanced chemical equation to show the concentration of ore in Baeyer’s process.
Aluminium hydroxide to alumina
The following sketch illustrates the process of conversion of Alumina to Aluminium:
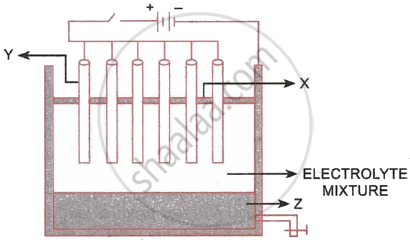
- Name the constituent of the electrolyte mixture which has a divalent metal in it.
- Name the powdered substances ‘X’ sprinkled on the surface of the electrolyte mixture.
- What is the name of the process?
- Write the reactions taking place at the electrodes ‘Y’ (anode) and ‘Z’ (cathode), respectively.
