Advertisements
Advertisements
प्रश्न
For the substance given below, describe the role played in the extraction of aluminium.
Sodium hydroxide
उत्तर
In the extraction of aluminium, sodium hydroxide plays two roles. First, finely grinded bauxite (ore of aluminium) is heated under pressure with conc. Caustic soda solution (NaOH solution) for 2-8 hours at 140°C to 150°C to produce sodium aluminate. The chemical equation is as follows:
\[\ce{Al2O3.2H2O + 2NaOH → 2NaAlO2 + 3H2O}\]
Second, when the sodium aluminate is diluted with water and cooled to 50°C, the sodium aluminate is hydrolysed to produce aluminium hydroxide as the precipitate. Here, the impurities dissolve in sodium hydroxide.
APPEARS IN
संबंधित प्रश्न
Write the equation for the reaction that occurs at the cathode during the extraction of aluminum by electrolysis.
Name the constituents of Brass.
Aluminium is extracted from its chief ore, bauxite. The ore is first purified and then the metal is extracted from it by electrolytic reduction.
Write an equation for the reaction which takes place at the anode during the extraction of aluminium by the electrolytic process.
Give reason for the following:
Extraction of aluminium was very difficult in the beginning.
How is aluminium obtained from pure ore?
Explain and give reasons why aluminium vessels should not be cleaned with powders containing alkalis.
In Hoope's process, pure aluminium is collected at the ______ of the electrolytic cell.
Name the following:
A metal which is always present in the amalgam.
State the relevant reason for the following:
A layer of powered coke is used over the electrolytic mixture in Hall Heroult’s process.
The following sketch illustrates the process of conversion of Alumina to Aluminium:
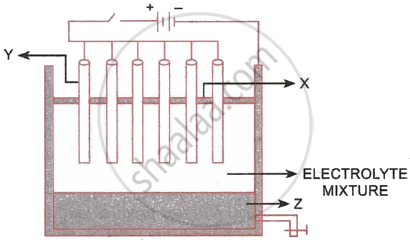
- Name the constituent of the electrolyte mixture which has a divalent metal in it.
- Name the powdered substances ‘X’ sprinkled on the surface of the electrolyte mixture.
- What is the name of the process?
- Write the reactions taking place at the electrodes ‘Y’ (anode) and ‘Z’ (cathode), respectively.
