Advertisements
Advertisements
प्रश्न
Give scientific reasons for the following:
Atomic radius goes on decreasing while going from left to right in a period.
उत्तर
- While going from left to right within a period, the atomic number increases one by one, meaning the positive charge on the nucleus increases by one unit at a time.
- However, the additional electron gets added to the same outermost shell. Due to the increased nuclear charge, the electrons are pulled towards the nucleus to a greater extent and thereby the size of the atom decreases.
APPEARS IN
संबंधित प्रश्न
Arrange the following as per the instruction given in the bracket:
Mg, Cl, Na, S, Si (decreasing order of atomic size).
Answer the following in respect of element `31/15 P`
Give its electronic configuration
Give the trends in atomic size on moving across the period left to right.
Arrange the elements of second and third periods in increasing order of their atomic size (excluding noble gases).
Why is the size of neon greater than fluorine?
Which is greater in size an atom or an anion?
Write scientific reason.
Atomic radius goes on increasing down a group.
Select the correct answer
Atomic radii of fluorine and neon in angstrom unit are
Arrange the following as per the instruction given in the bracket
Na, K, Li (Increasing atomic size)
The size of an atom is indicated by its _______.
Elements have been arranged in the following sequence on the basis of their increasing atomic masses.
| F, | Na, | Mg, | Al, | Si, | P, | S, | Cl, | Ar, | K |
- Pick two sets of elements which have similar properties.
- The given sequence represents which law of classification of elements?
Write the formula of the product formed when the element A (atomic number 19) combines with the element B (atomic number 17). Draw its electronic dot structure. What is the nature of the bond formed?
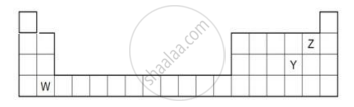
The diagram below shows part of the periodic table.
- Which elements would react together to form covalent compounds?
- Between the two elements W and Z, which will have a bigger atomic radius? Why?
An element with the largest atomic radius among the following is ______.
Arrange the following as per instruction given in the bracket.
Mg, Cl, Na, S, Si (decreasing order of atomic size)
Arrange the following in order of increasing radii:
Mg2+, Mg, Mg+
Explain your choice.
In Period 3 of the Periodic Table, element B is placed to the left of element A. On the basis of this information, choose the correct word from the option to complete the following statement:
The element A would have ______ atomic size than B.
Which one has the largest size?
Give reason for the following:
The size of a Cl− ion is greater than the size of a Cl atom.
