Advertisements
Advertisements
प्रश्न
Give the structure of the product you would expect when the following alcohol reacts with HCl–ZnCl2.
2-Methylbutan-2-ol
उत्तर
\[\begin{array}{cc}
\ce{CH3}\phantom{.................................}\ce{CH3}\phantom{..}\\
\phantom{.}|\phantom{.....................................}|\phantom{.....}\\
\ce{CH3 - C - CH2CH3 + HCl_{(conc.)}->[ZnCl2]CH3 - C - CH2CH3}\\
\phantom{.}|\phantom{.....................................}|\phantom{.....}\\
\phantom{..}\ce{\underset{2-methylbutane-2-ol}{OH}\phantom{.....................}\ce{\underset{2-chloro-2-methylbutane}{Cl}}}\phantom{..}\
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Write the equation involved in the acetylation of Salicylic acid.
Account for the following:
o-nitrophenol is more steam volatile than p-nitrophenol.
Intermolecular hydrogen bonding is strongest in ______.
Phenol is more acidic than alcohol because ____________.
Acidity of phenol is due to ____________.
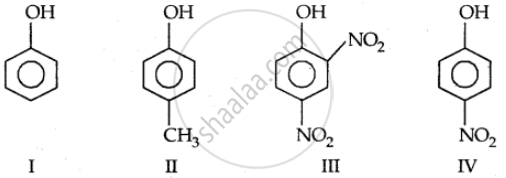
In the following compounds:
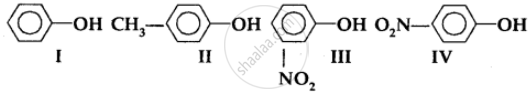
The order of acidity is
In CH3CH2OH, the bond that undergoes heterolytical change most readily is ____________.
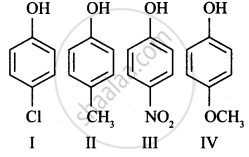
Strength of acidity is in order:
Arrange the following in decreasing order of acidic character:
What is the correct order of reactivity of alcohols in the following reaction?
\[\ce{R-OH + HCl ->[ZnCl2] R-Cl + H2O}\]
Mark the correct order of decreasing acid strength of the following compounds.
| (a) | 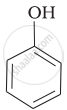 |
| (b) | 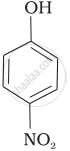 |
| (c) | 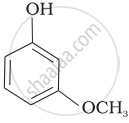 |
| (d) | 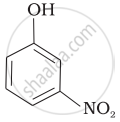 |
| (e) | 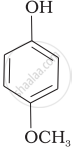 |
Out of o-nitrophenol and o-cresol which is more acidic?
For the pair phenol and cyclohexanol, answer the following:
Why is phenol more acidic than cyclohexanol?
Give the structure of the product you would expect when the following alcohol reacts with HBr.
Butan-1-ol
Give the structure of the product you would expect when the following alcohol reacts with SOCl2.
Butan-1-ol
Give the structure of the product you would expect when the following alcohol reacts with SOCl2.
2-Methylbutan-2-ol
