Advertisements
Advertisements
प्रश्न
Give the structures of A, B and C in the following reaction:
\[\ce{CH3COOH ->[NH3][\Delta] A ->[NaOBr] B ->[NaNO2/HCl] C}\]
उत्तर
\[\ce{\underset{Ethanoic acid}{CH3COOH} ->[NH3][\Delta] \underset{(A)}{\underset{Ethanamide}{CH3CONH2}} ->[NaOBr] \underset{(B)}{\underset{Methanamine}{CH3NH2}} ->[NaNO2/HCl] \underset{(C)}{\underset{Methanol}{CH3OH}}}\]
APPEARS IN
संबंधित प्रश्न
An aromatic compound 'A' of molecular formula C7H7ON undergoes a series of reactions as shown below. Write the structures of A, B, C, D and E in the following reactions :

Why cannot aromatic primary amines be prepared by Gabriel phthalimide synthesis?
Write the reactions of aromatic with nitrous acid.
Mendius reaction is used to convert _____________
Identify compound 'B' in following series of reactions?
\[\ce{Acetonitrile ->[Na/alcohol] A ->[NaNO2/dil.HCI] B}\]
Acetamide on reduction using Na/C2H5OH gives ____________.
Which of the following compounds is obtained when quaternary ammonium hydroxide is strongly heated?
Benzylamine may be alkylated as shown in the following equation:
\[\ce{C6H5CH2NH2 + R - X -> C6H5CH2NHR}\]
Which of the following alkylhalides is best suited for this reaction through SN1 mechanism?
Which of the following reagents would not be a good choice for reducing an aryl nitro compound to an amine?
How will you carry out the following conversion?
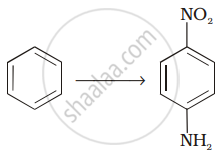
Assertion: Hoffmann’s bromamide reaction is given by primary amines.
Reason: Primary amines are more basic than secondary amines.
C6H5CONHCH3 can be converted into C6H5CH2NHCH3 by:-
Methyl amine on reaction with chloroform in the presence of NaOH gives ______.
- Phenyl methenamine
- N, N - Dimethylaniline
- N - Methyl aniline
- Benzenamine
Choose the correct order of the basic nature of the above amines.
Which of the following statement(s) is/are incorrect in case of Hofmann bromamide degradation?
Identify A and B in the following reaction.
\[\ce{C6H5CH2Br ->[Alco.][KCN] A ->[Na/Ethanol][reduction] B}\]
Write a short note on Ammonolysis.
Write short note on the following:
Ammonolysis
Write a short note on Ammonolysis.
Assertion: Amimonolysis of alkyl halides involves the reaction between alkyl halides and alcoholic ammonia.
Reason: Ammonolysis of alkyl halides produces secondary amines only.
