Advertisements
Advertisements
प्रश्न
Why cannot aromatic primary amines be prepared by Gabriel phthalimide synthesis?
उत्तर १
Gabriel phthalimide synthesis is used for the preparation of aliphatic primary amines. It involves nucleophilic substitution (SN2) of alkyl halides by the anion formed by the phthalimide.

But aryl halides do not undergo nucleophilic substitution with the anion formed by the phthalimide.
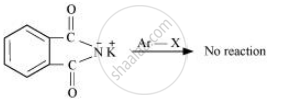
Hence, aromatic primary amines cannot be prepared by this process.
उत्तर २
The success of Gabriel phthalimide reaction depends upon the nucleophilic attack by the phthalimide anion on the organic halogen compound.
Since aryl halides do not undergo nucleophilic substitution reactions easily, therefore, arylamines, i.e., aromatic, primary amines cannot be prepared by Gabriel phthalimide reaction.

APPEARS IN
संबंधित प्रश्न
How is ethyl amine prepared from methyl iodide?
Identify the compounds 'A' and 'B' in the following equation:

Accomplish the following conversion:
Nitrobenzene to benzoic acid
Give the structures of A, B and C in the following reactions :
The following amines is the product of Gabriel phthalimide synthesis.
Mendius reaction is used to convert _____________
Identify compound 'B' in following series of reactions?
\[\ce{Acetonitrile ->[Na/alcohol] A ->[NaNO2/dil.HCI] B}\]
Which of the following amines exhibits maximum degree of intermolecular hydrogen bonding?
\[\ce{CH3-CN ->[Na/C2H5OH]}\]
The product formed is ____________.
Which of the following reactions is appropriate for converting benzamide to aniline?
Identify 'A' and 'B' in the following conversions.
\[\ce{CH3 - I ->[Alc. KCN][\Delta] A ->[Na/C2H5OH] B}\]
Which of the following reactions does NOT yield an amine?
For producing amines, the reaction of nitro compounds with iron scrap is preferred because:
Among the following amines, the strongest Brönsted base is:
What is the product when \[\ce{C6H5CH2NH2}\] reacts with \[\ce{HNO2}\]?
How will you carry out the following conversion?
A compound 'A' on reduction with iron scrap and hydrochloric acid gives compound 'B' with molecular formula C6H7N. Compound 'B' on reaction with CHCl3 and alcoholic KOH produces an obnoxious smell of carbylamine due to the formation of 'C'. Identify 'A', 'B' and 'C' and write the chemical reactions involved.
Which of the following would not be a good choice for reducing nitrobenzene to aniline?
Amides can be converted into amines by the reaction named ______.
Write short note on the following:
Ammonolysis
