Advertisements
Advertisements
प्रश्न
Give two important uses of baking soda.
उत्तर
Two important uses of baking soda are:
- It is mainly used in the preparation of baking powder. Baking powder is a mixture of baking soda and a mild acid known as tartaric acid. When it is heated or mixed in water, it releases CO2 that makes bread or cake fluffy.
- It is used in soda-acid fire extinguishers.
- It can be used to test the garden soil for acidity. If bubbles develop, then the soil Is too acidic
- If used on washing car, it will remove dead bug bodies without damaging the color or paint on the car.
- It is used in medicines as an antacid to remove the acidity of the stomach. Therefore, it is an important constituent of an antacid.
APPEARS IN
संबंधित प्रश्न
Write two applications of sodium bicarbonate (baking soda).
A student takes about 2 mL ethanoic acid in a dry test tube and adds a pinch of sodium hydrogen carbonate to it. He reports the following observations:
I. Immediately a colourless and odourless gas evolves with a brisk effervescence.
II. The gas turns lime water milky when passed through it.
III. The gas burns with an explosion when a burning splinter is brought near it.
IV. The gas extinguishes the burning splinter that is brought near it.
The correct observations are
(A) I, II and III
(B) II, III and IV
(C) III, IV and I
(D) I, II and IV
Name the substance which on treatment with chlorine yields bleaching powder?
Which compound is used as an antacid in medicine : NaHCO3 or Na2CO3?
Complete and balance the following chemical equations:
`NaHCO_3` 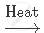
What is baking soda?
Write the chemical name of baking soda. Give the important uses of baking soda. How does baking soda differ chemically from washing soda?
Describe how sodium hydrogencarbonate (baking soda) is produced on a large scale. Write equation of the reaction involved.
Name two constituents of baking powder.
How does baking powder differ from baking soda?
Explain the action of baking powder in the making of cake (or bread). Write equation of the reaction involved.
In addition to sodium hydrogencarbonate, baking powders contain a substance X. Name the substance X. What is the role of substance X in the baking powder?
The formula of baking soda is:
(a) K2CO3
(b) KHCO3
(c) NaHCO3
(d) Na2CO3
The chemical name of baking soda is ______.
Write the chemical formula.
Baking soda
Salt 'P', commonly used in bakery products, on heating gets converted into another salt 'Q' which itself is used for the removal of hardness of water and a gas 'R' is evolved. The gas 'R', when passed through freshly prepared lime water, turns milky. Identify 'P', 'Q' and 'R', giving a chemical equation for the justification of your answer.
NaHCO, formed by reaction of ____________.
